Peer-reviewed publications
The olfactory bulb supports gamma oscillations in multiple target areas, involved in mood regulation. Depression can be alleviated by closed loop enhancement of gamma coupling between the bulb and target structures (in rodents, for now)
Observer/reader-dependent definition of synchrony and routing of information by phase of brain oscillations.
Cognition is environment-disengaged action.
40Hz flickering stimulation fail to entrain native gamma oscillations. Instead, visual neurons show sustained narrow-band responses, superimposed on native gamma.
- Inhibition dominates over excitation during theta oscillations in the CA1 region
- Concerted excitation/disinhibition favors place field properties in simulations
- DREADDs activation of interneurons impairs place field features in CA1
- A transient decrease of inhibition is critical for place field expression
In utero electroporation of plasmids for birthdating CA1 pyramidal neurons and dorsoventral occupancy of neurons as a function of birthdate.
Take home message: neuron born together will wire together and fire together.
Whether RF radiation can modulate ongoing neuronal activity by non-thermal mechanisms has been debated since the 1960s. To address this question, we monitored the neuronal activity of awake rodent brains under RF energy exposure (at 950 MHz) and in sham control paradigms. We modified a miniature 1-photon calcium imaging device to record interference-free neural activity and compared the results to those acquired using metal-containing silicon probes. Spiking activity was reliably affected by RF energy in metal-containing systems. However, we did not observe neuronal responses using metal-free optical recordings at induced local electric field strengths up to 230 V/m. Our results suggest that RF exposure higher than levels that are allowed by regulatory limits in real-life scenarios do not affect neuronal activity.

The current dominant view of the hippocampus is that it is a navigation “device” guided by environmental inputs. Yet, a critical aspect of navigation is a sequence of planned, coordinated actions. We examined the role of action in the neuronal organization of the hippocampus by training rats to jump a gap on a linear track. Recording local field potentials and ensembles of single units in the hippocampus, we found that jumping produced a stereotypic behavior associated with consistent electrophysiological patterns, including phase reset of theta oscillations, predictable global firing-rate changes, and population vector shifts of hippocampal neurons. A subset of neurons (“jump cells”) were systematically affected by the gap but only in one direction of travel. Novel place fields emerged and others were either boosted or attenuated by jumping, yet the theta spike phase versus animal position relationship remained unaltered. Thus, jumping involves an action plan for the animal to traverse the same route as without jumping, which is faithfully tracked by hippocampal neuronal activity.
During physiological conditions, brain temperature fluctuates approximately 3°C between sleep and active waking. Here, we show that features of hippocampal ripples, including the rate of occurrence, peak frequency, and duration are correlated with brain temperature variations. Focal bidirectional manipulation of temperature in the hippocampal CA1 region in awake rodents shows that ripple frequency can be altered in the direction expected from the correlational observations, implying that temperature plays a significant role.
Bidirectional communication between the hippocampus and other brain areas via sharp wave ripples (SPW-Rs) has been hypothesized to play an important role in cognitive functions. However, brain-wide coupling to SPW-Rs has been difficult to study at high temporal and spatial resolution. Here, we show that SPW-Rs coincide with transient brain-wide increase of functional connectivity. The diversity in SPW-R features was related to slower excitability fluctuations in cortical and subcortical areas. In return, SPW-Rs correlated with the timing, sign, and magnitude of partner structure responses. Our findings provide the most extensive survey of spiking activity surrounding hippocampal SPW-Rs to date.
Understanding how excitatory (E) and inhibitory (I) inputs are integrated by neurons requires monitoring their subthreshold behavior. We probed the subthreshold dynamics using optogenetic depolarizing pulses in hippocampal neuronal assemblies in freely moving mice.
By linking the past with the future, our memories define our sense of identity. Because human memory engages the conscious realm, its examination has historically been approached from language and introspection and proceeded largely along separate parallel paths in humans and other animals. Here, we first highlight the achievements and limitations of this mind-based approach and make the case for a new brain-based understanding of declarative memory with a focus on hippocampal physiology.
Highlights
- Unilateral or bilateral silencing of the mEC decreases theta and gamma currents
- Bilateral but not unilateral mEC silencing causes remapping in CA1 cell assemblies
- CA3 silencing decreases CA1 firing rates and leads to place cell remapping
- CA1 place cells and assemblies persist despite combined mEC and CA3 silencing
Highlights
- CA1 axo-axonic cells (AACs) are selectively labeled in Unc5b-2A-CreERT2 mice
- AACs are activated by locomotion or whisking in awake mice
- AACs exert inhibitory postsynaptic effects on CA1 pyramidal cells in vivo
- AAC optogenetic silencing induces novel place fields in pyramidal cells
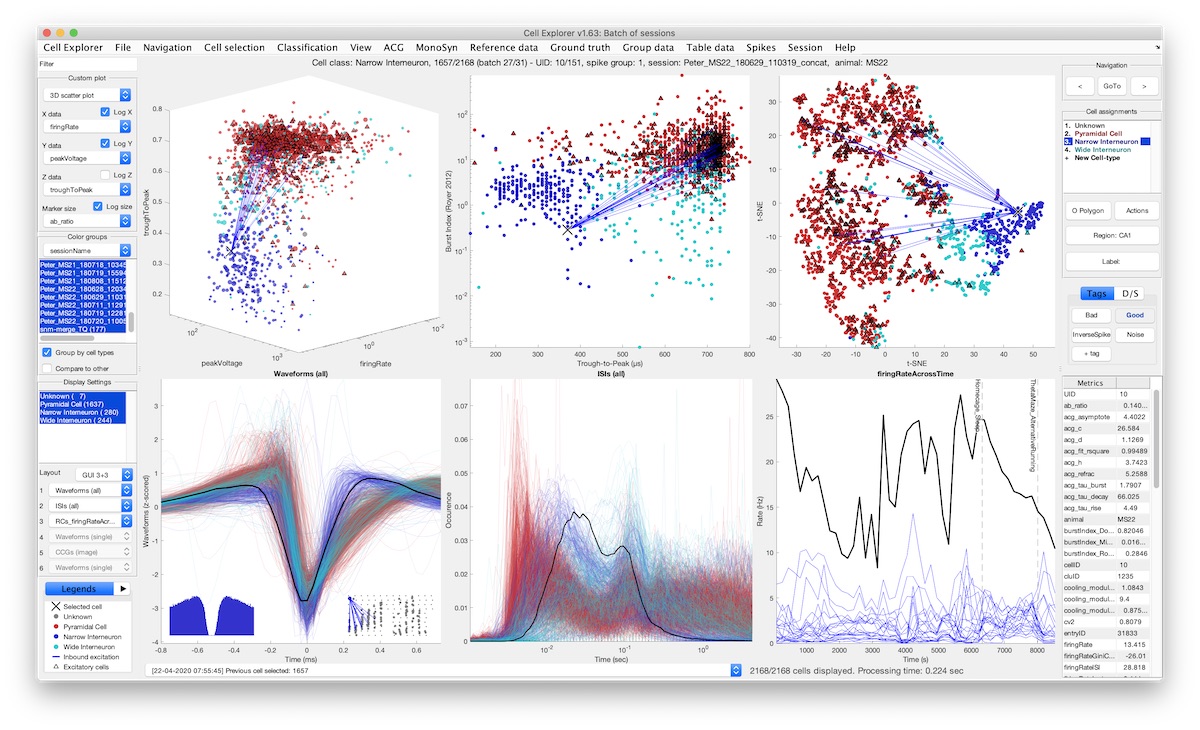
The large diversity of neuron types provides the means by which cortical circuits perform complex operations. Neurons can be described by biophysical and molecular characteristics, afferent inputs, and neuron targets. To quantify, visualize, and standardize those features, we developed the open-source, MATLAB-based framework CellExplorer. It consists of three components: a processing module, a flexible data structure, and a powerful graphical interface.
Highlights
- An open-source framework for single-cell characterization and visualization
- A processing module that calculates a set of standardized physiological metrics
- A graphical interface to explore computed features at the speed of a mouse click
Read the preview “Explorers of the cells: Toward cross-platform knowledge integration to evaluate neuronal function” by Liset de la Prida and Giorgio Ascoli
Mature neural networks synchronize and integrate spatiotemporal activity patterns to support cognition. Emergence of these activity patterns and functions is believed to be developmentally regulated, but the postnatal time course for neural networks to perform complex computations remains unknown. We investigate the progression of large-scale synaptic and cellular activity patterns across development using high spatiotemporal resolution in vivo electrophysiology in immature mice.
The hippocampus has previously been implicated in both cognitive and endocrine functions. We simultaneously measured electrophysiological activity from the hippocampus and interstitial glucose concentrations in the body of freely behaving rats to identify an activity pattern that may link these disparate functions of the hippocampus.

High-yield electrophysiological extracellular recording in freely moving rodents provides a unique window into the temporal dynamics of neural circuits. Recording from unrestrained animals is critical to investigate brain activity during natural behaviors. The use and implantation of high-channel-count silicon probes represent the largest cost and experimental complexity associated with such recordings making a recoverable and reusable system desirable. To address this, we have designed and tested a novel 3D printed head-gear system for freely moving mice and rats. The system consists of a recoverable microdrive printed in stainless steel and a plastic head cap system, allowing researchers to reuse the silicon probes with ease, decreasing the effective cost, and the experimental effort and complexity. The cap designs are modular and provide structural protection and electrical shielding to the implanted hardware and electronics. We provide detailed procedural instructions allowing researchers to adapt and flexibly modify the head-gear system.
Theta oscillations supported by the medial septum are believed to be a critical mechanism for learning and memory. We report that, in addition to theta oscillations, hippocampal SPW-Rs are important. Enhanced cholinergic activation in the hippocampus during the delay between choices in a spontaneous alternation task abolished SPW-Rs and impaired choice performance in mice. Our findings also demonstrate that the outcome of optogenetic manipulation of a key neurotransmitter, acetylcholine (ACh), depends on the state of the brain at the time of the perturbation.

Highlights
- μLED optogenetic stimulation induces remapping in CA1 neurons
- Stimulated and non-stimulated neurons show comparable place field reorganization
- Fields emerge in places with weak preexisting drive, not at site of depolarization
- Stimulation changes coupling between pyramidal cells and neighboring interneurons
Summary
Memory models often emphasize the need to encode novel patterns of neural activity imposed by sensory drive. Prior learning and innate architecture likely restrict neural plasticity, however. Here, we test how the incorporation of synthetic hippocampal signals is constrained by preexisting circuit dynamics. We optogenetically stimulated small groups of CA1 neurons as mice traversed a chosen segment of a linear track, mimicking the emergence of place fields. Stimulation induced persistent place field remapping in stimulated and non-stimulated neurons. The emergence of place fields could be predicted from sporadic firing in the new place field location and the temporal relationship to peer neurons before the optogenetic perturbation. Circuit modification was reflected by altered spike transmission between connected pyramidal cells and inhibitory interneurons, which persisted during post-experience sleep. We hypothesize that optogenetic perturbation unmasked sub-threshold place fields. Plasticity in recurrent/lateral inhibition may drive learning through the rapid association of existing states.
Pyramidal cells and GABAergic interneurons fire together in balanced cortical networks. In contrast to this general rule, we describe a distinct neuron type in mice and rats whose spiking activity is anti-correlated with all principal cells and interneurons in all brain states but, most prevalently, during the down state of non-REM (NREM) sleep.
Chemogenic activation of interneurons can bring about unexpected short-term and long-term changes in circuit dynamics. DREADD activation of all interneuron types suppressed firing of pyramidal cells but, unexpectedly, individual interneurons did not display sustained firing but instead undulated firing rates, interleaved with other interneurons. Despite the tonic suppression of spikes of individual pyramidal neurons, population bursts underlying sharp wave ripples persisted, often with stronger synchrony. Repeated drug application induced progressively weaker changes over days. Such plastic effects of DREADD activation should be taken into account in the interpretation of behavioral consequences.
The hippocampus is thought to guide navigation by forming a cognitive map of space. Different environments differ in geometry and the availability of cues that can be used for navigation. Although several spatial coding mechanisms are known to coexist in the hippocampus, how they are influenced by various environmental features is not well understood. To address this issue, we examined the spatial coding characteristics of hippocampal neurons in mice and rats navigating in different environments. We found that CA1 place cells located in the superficial sublayer were more active in cue-poor environments and preferentially used a firing rate code driven by intra-hippocampal inputs. In contrast, place cells located in the deep sublayer were more active in cue-rich environments and used a phase code driven by entorhinal inputs. Switching between these two spatial coding modes was supported by the interaction between excitatory gamma inputs and local inhibition.

In 2001, I was invited to write a review for a prominent journal. I thought that the best way to exploit this opportunity was to write an essay about my problems with ill-defined scientific terms and question whether the dominant framework in neuroscience is on the right track. My main argument was that many terms in neuroscience are inherited from folk psychology and are often used in two ambiguous ways: both as the thing-to-be-explained (explanandum) and the thing-that-explains (explanans; e.g., “we have memory because we remember,” “we remember because we have memory”). These postulated terms are assumed to be entities with definable boundaries, and within this framework, the goal of neuroscience is to find homes and mechanisms for these terms in the brain with corresponding boundaries (I called this “the correlational approach”). I warned that a framework dictated by human-centric introspection might not be the right roadmap for neuroscience and argued that there should be another way of carving up the brain’s “natural kinds.”
Petersen et al. demonstrate that cooling of the medial septum slows theta oscillation and increases choice errors without affecting spatial features of pyramidal neurons. Cooling affects distance-time, but not distance-theta phase, compression. The findings reveal that cell assemblies are organized by theta phase and not by external (clock) time.
The combination of in vivo extracellular recording and genetic-engineering-assisted optical stimulation is a powerful tool for the study of neuronal circuits. Precise analysis of complex neural circuits requires high-density integration of multiple cellular-size light sources and recording electrodes. However, high-density integration inevitably introduces stimulation artifact. We present minimal-stimulation-artifact (miniSTAR) μLED optoelectrodes that enable effective elimination of stimulation artifact. A multi-metal-layer structure with a shielding layer effectively suppresses capacitive coupling of stimulation signals. A heavily boron-doped silicon substrate silences the photovoltaic effect induced from LED illumination.
We demonstrate that this coupling is brain state-dependent, and delineate a topographically-organized anatomical pathway via VGlut2-expressing, bursty neurons in the subiculum. Optogenetic stimulation or inhibition of bursty subicular cells induced or reduced responses in superficial gRSC, respectively. These results identify a specific path and underlying mechanisms by which the hippocampus can convey neuronal content to the neocortex during SPW-Rs.
We hypothesize that SWRs act as a multiplexed signal, the mnemonic specificity of which is largely determined by this transformation function and discuss the implications of this framing for theories of systems consolidation.
The hippocampus is heavily studied in the context of spatial navigation, and the format of spatial information in the hippocampus is multifaceted and complex. Furthermore, the hippocampus is also thought to contain information about other important aspects of behavior such as running speed, although there is not agreement on the nature and magnitude of their effect. To understand how all of these variables are simultaneously represented and used to guide behavior, a theoretical framework is needed that can be directly applied to the data we record. We present a model that captures well-established spatial-encoding features of hippocampal activity and provides the opportunity to identify and incorporate novel features for our collective understanding.
Highlights
- Lateral septal neurons can synchronize to produce local high-frequency oscillations
- Lateral septal high-frequency oscillations are coupled to hippocampal ripples
- The magnitude, but not content, of hippocampal ripples are read out by lateral septum
Prior studies have demonstrated that a closed-loop acoustic pulse paradigm during sleep can enhance verbal memory performance. This technique has widespread scientific and clinical appeal due to its noninvasive nature and ease of application. We tested with a rigorous double-blind design whether this technique could enhance key sleep rhythms associated with sleep-dependent memory performance. We discovered that we could reliably enhance slow and spindle rhythms, but did not improve memory performance in the stimulation condition compared with sham condition. Our findings suggest that enhancing slow-spindle rhythms is insufficient to enhance sleep-dependent learning.

What started as a self-scrutinizing exercise turned into an extraordinary volume on a journey in search of the origin of survival mechanisms and consciousness. Joseph LeDoux — the undisputed guru of emotions and the Henry and Lucy Moses Professor of Science at New York University — declares in The Deep History of Ourselves that the notion of an ‘amygdala fear center’ that became not only a scientific doctrine but also a cultural meme is dead wrong. Since his writings contributed to this mischaracterization, LeDoux feels that he has some explaining to do. He does this in the form of beautifully composed prose of 400 pages. Without hesitation, he tells the reader up front that, in order to accomplish his goals, a “radical approach is needed”. This is done in the form of two interconnected journeys, the first of which is the evolution of survival circuits: “there is indeed good evidence that the same brain systems control survival behaviors in humans and other mammals”. However, these are not the systems that are responsible for the conscious feelings that we experience when we engage in such behaviors and, therefore, he postulates the need of another system that is uniquely human. To contrast these two lines of ideas, the author reaches back through four billion years of natural history. The elaborate discussion on our long past is done not for the sake of cataloguing facts but to seek answers to a complex problem: the origin of our phenomenal or access consciousness. In the process, two new LeDouxs unfold: LeDoux the evolutionary biologist, evo–devo guy and a consciousness expert.
Preview
Using clever experimental design and exploiting the high temporal resolution power of magnetoencephalography, Liu et al. show in humans how “offline” reactivation of brain patterns allows the abstraction of new knowledge from previous experience. The key mechanism may involve hippocampal sharp-wave ripples.
Sharp wave ripples in the hippocampus are thought to play a role in memory formation and action planning. Fernández-Ruiz et al. used multisite electrophysiological recordings combined with optogenetic activation of hippocampal pyramidal neurons in rats performing learning tasks. Learning and correct recall in spatial memory tasks were associated with extended sharp wave ripples. Artificially prolonging these ripples improved working memory performance, whereas aborting the late part of ripples decreased performance.
During non-rapid eye movement (NREM) sleep, neuronal populations in the mammalian forebrain alternate between periods of spiking and inactivity. Termed the slow oscillation in the neocortex and sharp wave-ripples in the hippocampus, these alternations are often considered separately but are both crucial for NREM functions. By directly comparing experimental observations of naturally-sleeping rats with a mean field model of an adapting, recurrent neuronal population, we find that the neocortical alternations reflect a dynamical regime in which a stable active state is interrupted by transient inactive states (slow waves) while the hippocampal alternations reflect a stable inactive state interrupted by transient active states (sharp waves).
Highlights
- Multisite LFP recording and LFP-spike coupling identified physiological layers in V1
- The prominent 3–6 Hz LFP shared characteristic features with primate alpha rhythm
- Spike transmission strength from layer 2/3 to layer 5 neurons was stronger during waking
- A subset of layer 6 neurons was active selectively in the DOWN state of non-REM sleep
Here, we review data and modelling on the immediate neurophysiological effects of TES in vitro as well as in vivo in both humans and other animals. While it remains unclear how typical TES protocols affect neural activity, we propose that validated models of current flow should inform study design and artifacts should be carefully excluded during signal recording and analysis. Potential indirect effects of TES (e.g., peripheral stimulation) should be investigated in more detail and further explored in experimental designs. We also consider how novel technologies may stimulate the next generation of TES experiments and devices, thus enhancing validity, specificity, and reproducibility.
We propose that the hippocampus performs a general but singular algorithm: producing sequential content-free structure to access and organize sensory experiences distributed across cortical modules.
Neural ‘representations’ can be referenced to many frames, and direct comparisons across frames of reference can be used when trying to identify underlying neural computations.
Neural dynamics and transformations can be described without resorting to the concepts of space and time.
Future research should focus on transformation rules between structures, rather than on tuning. A neuronal observer-centered approach that compares two internal variables may be more fruitful than correlating an external signal with neuronal patterns.
The hippocampus constructs a map of the environment. How this “cognitive map” is utilized by other brain regions to guide behavior remains unexplored. To examine how neuronal firing patterns in the hippocampus are transmitted and transformed, we recorded neurons in its principal subcortical target, the lateral septum (LS). We observed that LS neurons carry reliable spatial information in the phase of action potentials, relative to hippocampal theta oscillations, while the firing rates of LS neurons remained uninformative.
Conditioned place preference (CPP) is a widely used model of addiction-related behavior whose underlying mechanisms are not understood. In this study, we used dual site silicon probe recordings in freely moving mice to examine interactions between the hippocampus and nucleus accumbens in cocaine CPP. We found that CPP was associated with recruitment of D2-positive nucleus accumbens medium spiny neurons to fire in the cocaine-paired location, and this recruitment was driven predominantly by selective strengthening of coupling with hippocampal place cells that encode the cocaine-paired location.
Highlights
- Cocaine place conditioning increases subsequent firing in D2R medium spiny neurons
- Accumbens neurons decode spatial information from hippocampal inputs
- Increased accumbens firing is due to location-selective coupling with hippocampus
Optogenetics allows for optical manipulation of neuronal activity and has been increasingly combined with intracellular and extracellular electrophysiological recordings. Genetically-identified classes of neurons are optically manipulated, though the versatility of optogenetics would be increased if independent control of distinct neural populations could be achieved on a sufficient spatial and temporal resolution. We report a scalable multisite optoelectrode design that allows simultaneous optogenetic control of two spatially intermingled neuronal populations in vivo. We describe the design, fabrication, and assembly of low-noise, multisite/multicolor optoelectrodes.
Electrical interfacing with neural tissue is key to advancing diagnosis and therapies for neurological disorders, as well as providing detailed information about neural signals. A challenge for creating long-term stable interfaces between electronics and neural tissue is the huge mechanical mismatch between the systems. So far, materials and fabrication processes have restricted the development of soft electrode grids able to combine high performance, long-term stability, and high electrode density, aspects all essential for neural interfacing. Here, this challenge is addressed by developing a soft, high-density, stretchable electrode grid based on an inert, high-performance composite material comprising gold-coated titanium dioxide nanowires embedded in a silicone matrix.
Transcranial electric stimulation is a non-invasive tool that can influence brain activity; however, the parameters necessary to affect local circuits in vivo remain to be explored. Here, we report that in rodents and human cadaver brains, ~75% of scalp-applied currents are attenuated by soft tissue and skull.
The local field potential (LFP) is an aggregate measure of group neuronal activity and is often correlated with the action potentials of single neurons. In recent years, investigators have found that action potential firing rates increase during elevations in power high-frequency band oscillations (50-200 Hz range). However, action potentials also contribute to the LFP signal itself, making the spike-LFP relationship complex. Here, we examine the relationship between spike rates and LFP in varying frequency bands in rat neocortical recordings.
Animals integrate multiple sensory inputs to successfully navigate in their environments. Head direction (HD), boundary vector, grid and place cells in the entorhinal-hippocampal network form the brain’s navigational system that allows to identify the animal’s current location, but how the functions of these specialized neuron types are acquired remain to be understood. Here we report that activity of HD neurons is influenced by the ambulatory constraints imposed upon the animal by the boundaries of the explored environment, leading to spurious spatial information. However, in the post-subiculum, the main cortical stage of HD signal processing, HD neurons convey true spatial information in the form of border modulated activity through the integration of additional sensory modalities relative to egocentric position, unlike their driving thalamic inputs. These findings demonstrate how the combination of HD and egocentric information can be transduced into a spatial code.
The hippocampus serves a critical function in memory, navigation, and cognition. Nature Neuroscience asked John Lisman to lead a group of researchers in a dialog on shared and distinct viewpoints on the hippocampus.

Transcranial electrical stimulation has widespread clinical and research applications, yet its effect on ongoing neural activity in humans is not well established. Previous reports argue that transcranial alternating current stimulation (tACS) can entrain and enhance neural rhythms related to memory, but the evidence from non-invasive recordings has remained inconclusive. Here, we measure endogenous spindle and theta activity intracranially in humans during low-frequency tACS and find no stable entrainment of spindle power during non-REM sleep, nor of theta power during resting wakefulness. As positive controls, we find robust entrainment of spindle activity to endogenous slow-wave activity in 66% of electrodes as well as entrainment to rhythmic noise-burst acoustic stimulation in 14% of electrodes. We conclude that low-frequency tACS at common stimulation intensities neither acutely modulates spindle activity during sleep nor theta activity during waking rest, likely because of the attenuated electrical fields reaching the cortical surface.
Nothing is more intuitive, yet more complex, than the concepts of space and time. In contrast to spacetime in physics, space and time in neuroscience remain separate coordinates to which we attach our observations. Investigators of navigation and memory relate neuronal activity to position, distance, time point, and duration and compare these parameters to units of measuring instruments. Although spatial-temporal sequences of brain activity often correlate with distance and duration measures, these correlations may not correspond to neuronal representations of space or time. Neither instruments nor brains sense space or time. Neuronal activity can be described as a succession of events without resorting to the concepts of space or time. Instead of searching for brain representations of our preconceived ideas, we suggest investigating how brain mechanisms give rise to inferential, model-building explanations.
The consolidation of context-dependent emotional memory requires communication between the hippocampus and the basolateral amygdala (BLA), but the mechanisms of this process are unknown. We recorded neuronal ensembles in the hippocampus and BLA while rats learned the location of an aversive air puff on a linear track, as well as during sleep before and after training. We found coordinated reactivations between the hippocampus and the BLA during non-REM sleep following training. These reactivations peaked during hippocampal sharp wave–ripples (SPW-Rs) and involved a subgroup of BLA cells positively modulated during hippocampal SPW-Rs. Notably, reactivation was stronger for the hippocampus–BLA correlation patterns representing the run direction that involved the air puff than for the ‘safe’ direction. These findings suggest that consolidation of contextual emotional memory occurs during ripple-reactivation of hippocampus–amygdala circuits.
Explicit memory formation involves the transfer of rapidly encoded information from the hippocampus to long-term storage sites in the association cortex. Khodagholy et al. developed a microelectrode system for large-scale simultaneous electrophysiological monitoring of multiple sites in the rat neocortex. They observed discrete high-frequency neocortical oscillations called ripples only in the association cortex. These cortical ripples shared many properties with hippocampal ripples. Hippocampal ripples were coupled with cortical ripples in the posterior parietal cortex, an association cortical area linked to navigational planning. This coupling was increased during sleep after the induction of long-term hippocampal-dependent spatial memory.
English, McKenzie, et al. identify, validate, and quantify monosynaptic connections between pyramidal cells and interneurons, using the spike timing of pre- and postsynaptic neurons in vivo. Their large-scale method uncovers a backbone of connectivity rules in the hippocampus CA1 circuit.
Highlights
- Pyramidal cell-interneuron monosynaptic connections identified using spike timing
- Skewed distribution of connection probability and strength
- Short-term plasticity of connection strength is synapse specific
- Presynaptic cooperativity and postsynaptic timing impact spike transmission probability
Consolidation of memories require SPW-Rs. Since place cells carry such information in case of spatial memories, we asked whether place cells also benefit from SWP-Rs. We used closed-loop optogenetic methods to silence a subset of place cells near the reward locations selectively during SPW-R. We demonstrate that interference with SPW-R-associated activity during learning prevents stabilization and refinement of hippocampal spatial maps..
Sleep is thought to mediate both mnemonic and homeostatic functions. But which patterns of sleep are critical is less known. By combining empirical data and modeling, we suggest a mechanism by which spiking activity during the slow oscillation acts to maintain network statistics that promote a skewed distribution of neuronal firing rates. Perturbation of that activity by hippocampal replay acts to integrate new memory traces into the existing cortical network.

Theta-gamma phase coupling and spike timing within theta oscillations are prominent features of the hippocampus and are often related to navigation and memory. However, the mechanisms that give rise to these relationships are not well understood. Using high spatial resolution electrophysiology, we investigated the influence of CA3 and entorhinal inputs on the timing of CA1 neurons. The theta-phase preference and excitatory strength of the afferent CA3 and entorhinal inputs effectively timed the principal neuron activity, as well as regulated distinct CA1 interneuron populations in multiple tasks and behavioral states. Feedback potentiation of distal dendritic inhibition by CA1 place cells attenuated the excitatory entorhinal input at place field entry, coupled with feedback depression of proximal dendritic and perisomatic inhibition, allowing the CA3 input to gain control toward the exit. Thus, upstream inputs interact with local mechanisms to determine theta-phase timing of hippocampal neurons to support memory and spatial navigation.
The hippocampal dentate gyrus is often viewed as a segregator of upstream information. Physiological support for such function has been hampered by a lack of well-defined characteristics that can identify granule cells and mossy cells. We developed an electrophysiology-based classification of dentate granule cells and mossy cells in mice that we validated by optogenetic tagging of mossy cells. Granule cells exhibited sparse firing, had a single place field, and showed only modest changes when the mouse was tested in different mazes in the same room. In contrast, mossy cells were more active, had multiple place fields and showed stronger remapping of place fields under the same conditions. Although the granule cell-mossy cell synapse was strong and facilitating, mossy cells rarely “inherited” place fields from single granule cells. Our findings suggest that the granule cells and mossy cells could be modulated separately and their joint action may be critical for pattern separation.
Using large-scale recordings and large data bases, we describe the physiological properties and behavioral correlates of pyramidal cells in the CA3, CA2, and CA1 subregions and subnetworks. work cooperatively during spatial navigation and memory. These results indicate that a combination of intrinsic properties together with distinct intra- and extra-hippocampal inputs may account for the subregion-specific modulation of spiking dynamics and spatial tuning of neurons during behavior. This is a collaborative work with the Berenyi lab (http://www.berenyilab.com/).
This study demonstrates that sharp-wave ripples (SPW-Rs) in the hippocampus are induced most often by the strongly recurrent CA2 region. Synchronous activity of CA2 ensembles preceded SPW-R-related population activity in CA3 and CA1 regions. The trigger role of the CA2 region in SPW-R is more pronounced during waking than sleeping. This is a collaborative work with the Berenyi lab (http://www.berenyilab.com/)
We have advanced our first version of the NeuroGrid. Several different and larger spatial coverage architectures were tested with the assistance of our collaborators at both NYU and University of Iowa. In addition to anesthetized patients, we were able to record during surgery from a waking patient as well.
This collaborative work yielded an important improvement in optogenetic methods. We introduce a novel silicon probe with a monolithically integrated optical waveguide mixer that can deliver multicolor light at a common waveguide port to achieve multicolor modulation. This fully-integrated approach demonstrates the spatial precision and scalability needed to enable independent activation and silencing of the same or different groups of neurons while simultaneously recording from them.
Progress in spike sorting. A collaborative work with the Harris Lab, sparked by an idea of a curious CalTech summer student at Rutgers (J Schulman) a few years ago.
Firing rate distribution are more than just rate differences. Members at the ends of the lognormal spectrum have many different properties from how they are embedded in population events, connectivity, etc. Here we show that high firing rate hippocampal neurons form a pre-existing backbone of circuit dynamics (‘rigid’ neurons, blue) while it is the slower firing majority which learns effectively in novel situations (‘plastic’, red). These two ends of the continuous distributions appear to be responsible for capturing the familiar and novel aspects of any experience.
We describe here a new form of regulation of neural activity by sleep. The firing rates of fast- and slow-firing cortical neurons of the lognormal distribution are homogenized over sleep. This effect is due to contributions from REM, nonREM, and sleep microarousals.
Ripples, spindles and slow oscillations not only offer good things to the brain but their pathological coupling may have serious cognitive consequences. We show here that in temporal lobe epilepsy that spontaneous hippocampal epileptic discharges displace ripples, strongly couple to prefrontal spindles in both rats and humans and deteriorate memory. Next task: decouple these events in attempts to restore function.
Finally, silicon probes yield high quality signals from the monkey brain – and at a low cost!
This review is a result of a lunch conversation at the Hungarian Academy of Sciences in January 2016. Coming from very different conceptual directions we settled on some common computational principles.
Focal light delivery (nanoWatts amount) can selectively stimulate neuron members of local circuits; a great tool for analyzing local circuit operations.
We make all our data available for other neuroscientists for further exploration (http://www.nwb.org/). To download hippocampal, neocortical and thalamic datasets, see CRCNS.org.
This monograph is a A to Z text about the mechanisms and function of hippocampal sharp wave ripples.
We compared multi-neuronal spiking during theta oscillations, spontaneous ripples, and focal optogenetically induced high-frequency oscillations (“synthetic” ripples) in freely moving mice. Unexpectedly, firing rates and rate modulations of individual neurons, and multineuronal sequences of pyramidal cell and interneuron spiking were correlated across all conditions. Thus, participation of single neurons and their sequential order in population events are controlled by a combination of extrinsic inputs, local-circuit properties and single-neuron biophysics.
This review is tailored to non-neuroscientists using simple language.
Despite decades of extracellular action potential (EAP) recordings monitoring brain activity, the biophysical origin and inherent variability of these signals remains enigmatic. Here we performed whole-cell patch recordings of excitatory and inhibitory neurons in rat somatosensory cortex slice while positioning a silicon probe in their vicinity to concurrently record intra- and extracellular voltages for spike frequencies under 20 Hz. Using this approach, we characterize biophysical events and properties (intracellular spiking, extracellular resistivity, temporal jitter, etc.) related to EAP-recordings at the single-neuron level in a layer-specific manner.
Brain-implanted devices are no longer a futuristic idea. Herein, we discuss how advances in experimental closed-loop systems hold promise for improved clinical benefit in patients with neurological disorders.
The head-direction (HD) system is as fundamental as place cells and grid cells for spatial navigation. Similar to place cells and grid cells, HD neurons have been assumed to be ‘driven’ by peripheral sensors. While the role of environmental signals on assisting spatial navigation is unquestionable, neurons, circuits and systems involved in navigation remain coherent during ‘mental travel’ and sleep. This papers show that HD neurons in the antero-dorsal thalamic nucleus and the post-subiculum of mice sustain their temporal correlation structure during sleep. The findings provide experimental support for attractor networks in the brain.
This Primer explains the necessary steps for large-scale recordings of neuronal spiking combined with localized optogenetic perturbation for circuit probing. Closed-loop optogenetic stimulation of single or a few neurons is possible with a new generation of silicon probes using nanoW power of light emitted by microLEDs.
Here we propose a neuroscience-based model of cognition. Our core hypothesis is that cognition depends on internal models of the animal and its world, where internally generated sequences can serve to perform “what if” scenarios and anticipate the possible consequences of alternative actions without actually testing them, and aid in the decisions of overt actions. We support our hypotheses by several examples of recent experimental findings and show how externally guided cell assembly sequences become internalized to support cognitive functions.
Saul Steinberg’s cartoon ‘View of the World from 9th Avenue’ perfectly captures how neuronal populations of the hippocampus code for space. The majority of neurons have single place fields while a small fraction has many. In increasing size environments or in different rooms, new neurons are recruited but a minority of neurons are active at multiple locations. But there is a trick: the diligent minority fires more spikes and, at the end, may emit as many spikes as the the majority of slow firing neurons with single fields. Such skewed (log) statistics are typical at many levels of brain dynamics. Such representation may serve to combine ‘uniquely different’ and ‘familiar’ features.
Gamma, high gamma, super fast gamma. Do these terms correspond true oscillations and do they share similar mechanisms? Can they be used effectively and similarly for interregional communication? This Perspective discusses how electrode placement, spatial resolution and several other technical issues can affect the interpretation of results. It also provides a set of recommendation for appropriate interpretation of experimental results.
We developed an organic material-based, biocompatible, ultra-conformable, scalable neural interface array (the ‘NeuroGrid’) that can record both LFP and action potentials from superficial neurons without penetrating the brain surface. Spikes with features of interneurons and pyramidal cells were simultaneously acquired by multiple neighboring electrodes of the NeuroGrid, allowing for isolation of putative single neurons in rats. We also recorded LFP-modulated spiking activity intra-operatively in patients undergoing epilepsy surgery. The NeuroGrid constitutes an effective method for large-scale, stable recording of neuronal spikes in concert with local population synaptic activity, enhancing comprehension of neural processes across spatiotemporal scales and potentially facilitating diagnosis and therapy for brain disorders.
We took the challenge of figuring out how to perform simultaneously extracellular and intracellular data in freely behaving mice. In turn, this innovation allowed us to examine how population activity during sharp wave ripples is reflected by the membrane potential fluctuations of hippocampal pyramidal cells. We uncovered a strong shunting inhibition and demonstrate that spikes are initiated orthodromically due to transient gain of excitation over inhibition. These observations reveal important differences between findings in vitro and in vivo.
These findings surprised us! We found a sizable fraction of neuron pairs (mainly interneurons) with zero millisecond synchrony in both CA1 and CA1 hippocampal regions. This millisecond timescale synchrony appeared to be independent of network state, excitatory input, and oscillations and it can emerge independent of gap junctions. Importantly, we show that this synchronous spiking may cause stronger inhibition and rebound spiking in target neurons. Bottom line: zero time synchrony can be achieved with synaptic coupling across interneurons.
These experiments examined how the CA3 and entorhinal inputs affect the activity in the CA1 network through theta-gamma coupling. It shows that upstream neurons fire coherently with LFP gamma activity in the target dendritic layer. However, gamma coupling of principal cells across regions is rather poor and decreases with gamma frequency. LFP gamma activity in the somatic layer is a combination of passive return currents from the dendrites, feed-forward somatic inhibition and superimposed spikes of synchronously firing neurons.
Synaptic inhibition, brought about by a rich variety of interneuron types, counters excitation, modulates the gain, timing, tuning, bursting properties of principal cell firing, and exerts selective filtering of synaptic excitation. At the network level, it allows for coordinating transient interactions among the principal cells to form cooperative assemblies for efficient transmission of information and routing of excitatory activity across networks, typically in the form of brain oscillations. Recent techniques based on targeted expression of neuronal activity modulators, such as optogenetics, allow physiological identification and perturbation of specific interneuron subtypes in the intact brain. Combined with large-scale recordings or imaging techniques, these approaches facilitate our understanding of the multiple roles of inhibitory interneurons in shaping circuit functions.
The role of cholinergic inputs in hippocampal activity has been studied for decades. In this optogenetic study we should that activation of septal cholinergic neurons suppresses ripples, increases theta power under anesthesia but much less so during waking. Peritheta frequencies are suppress under both conditions. Images shows cholinergic neurons and fibers in medial septum.
These free data sets contain >25 hours of large-scale recordings of neuronal activity in the hippocampus and entorhinal cortex. We offer these data for public use with the understanding that data mining by bright people can result in new discoveries.
The mechanisms by which ripple oscillations contribute to memory consolidation are unclear. Using multisite closed-loop optogenetic manipulations in freely moving mice and rats, complemented with local pharmacological manipulation, we show that temporally precise local interactions between excitatory and inhibitory neurons underlie ripple generation. These findings constrain the mechanism involved in generation and spatial coordination of ripple oscillations.
Temporally evolving cell assemblies broadcast their spike outputs to target neurons in the form of EPSPs and IPSPs (‘synapsembles’). The induced transmembrane changes can also be measured in the extracellular space as spatio-temporally varying mesoscopic local field potentials (LFP). Monitoring large number of sites in the neuronal space therefore should offer rich information about the workings of the network. We tested this hypothesis in this work and demonstrate that the position of the rat in the environment can be reliably ‘read out’ from the ‘demodulated’ LFP which rides on the carrier theta oscillations in the hippocampus.
We often assume that the variables of functional and structural brain parameters — such as synaptic weights, the firing rates of individual neurons, the synchronous discharge of neural populations, the number of synaptic contacts between neurons and the size of dendritic boutons — have a bell-shaped distribution. However, at many physiological and anatomical levels in the brain, the distribution of numerous parameters is in fact strongly skewed with a heavy tail, suggesting that skewed (typically lognormal) distributions are fundamental to structural and functional brain organization.
The combination of optogenetics and large-scale neuronal recordings allows specific interneuron populations to be identified and perturbed for circuit analysis in intact animals. A crucial aspect of this approach is coupling electrophysiological recording with spatially and temporally precise light delivery. Recent progress demonstrates the utility and power of this novel technique for interneuron research.
Since the anatomical substrate that generate theta currents and spindle oscillation related currents in the hippocampus are very similar, we compared their physiological mechanisms. Despite the similarity of the mesoscopic LFP patterns, we find that the spike content organization of theta waves and spindle waves are dramatically different. Phase-organized metachrony characterizes theta oscillations, while neurons are largely synchronous during spindles.
A topographical relationship exists between the septotemporal segments of the hippocampus and their entorhinal–neocortical targets, but the physiological organization of activity along the septotemporal axis is poorly understood. We recorded sharp-wave ripple patterns in rats during sleep from the entire septotemporal axis of the CA1 pyramidal layer.
Despite the several-thousand-fold increase of brain volume during the course of mammalian evolution, the
hierarchy of brain oscillations remains remarkably preserved, allowing for multiple-time-scale communication within and across neuronal networks at approximately the same speed, irrespective of brain size.
Deployment of large-diameter axons of long-range neurons could be a key factor in the preserved time management in growing brains. We discuss the consequences of such preserved network constellation in mental disease, drug discovery, and interventional therapies.
Using optogenetic methods to activate parvalbumin immunoreactive interneurons (PV) in the hippocampus and neocortex we show that PV neurons can induce theta-band-limited, excess postinhibitory spiking in pyramidal cells (theta resonance).
Monitoring representative fractions of neurons from multiple brain circuits in behaving animals is necessary for understanding neuronal computation. Here we describe a system that allows high channel count recordings from a small volume of neuronal tissue using a lightweight signal multiplexing head-stage that permits free behavior of small rodents. These methods will allow the investigation of circuit operations and behavior-dependent inter-regional interactions for testing hypotheses of neural networks and brain function.
Using simultaneous recordings from multiple layers of the entorhinal–hippocampal loop, we found that coactivation of principal cell pairs (synchrony) was lowest during exploration and rapid-eye-movement (REM) sleep, associated with theta oscillations, and highest in slow wave sleep. Individual principal neurons had a wide range of theta phase preference. Thus, while theta oscillations reduce population synchrony, they nevertheless coordinate the phase (temporal) distribution of neurons. As a result, multiple cell assemblies can nest within the period of the theta cycle.
In many applications it is critical that only those neurons are activated and silenced which at the same time are monitored. In this work we demonstrate how a wave guide can be monolithically integrated silicon probes. High spatial precision enables optical excitation of virally or genetically targeted neurons with minimal power and recording of single-units in dense cortical and subcortical regions. Shown here is an 8-site silicon probe with an optical guide. Ideally, all future silicon probes should be equipped with wave guides described here.
This work addressed a long-standing ‘neuromyth’ (SFN Neuromyths; “We use only 10% of our brain”). The answers are both complex and interesting. They demonstrate that the brain is largely a preconfigured network where a minority of neurons does most of the work all the time. The lognormal distribution of firing rates, synaptic strengths and the magnitude of population patterns are likely a general rule and has important implications from perception (Weber law) to memory.
Here we propose that mechanisms of memory and planning have evolved from mechanisms of navigation in the physical world and hypothesize that the neuronal algorithms underlying navigation in real and mental space are fundamentally the same. We review experimental data in support of this hypothesis and discuss how specific firing patterns and oscillatory dynamics in the entorhinal cortex and hippocampus can support both navigation and memory.

The perpetual activity of the cerebral cortex is largely supported by the variety of oscillations the brain generates, spanning a number of frequencies and anatomical locations, as well as behavioral correlates. First, we review findings from animal studies showing that most forms of brain rhythms are inhibition-based, producing rhythmic volleys of inhibitory inputs to principal cell populations, thereby providing alternating temporal windows of relatively reduced and enhanced excitability in neuronal networks. These inhibition-based mechanisms offer natural temporal frames to group or “chunk” neuronal activity into cell assemblies and sequences of assemblies, with more complex multi-oscillation interactions creating syntactical rules for the effective exchange of information among cortical networks. We then review recent studies in human psychiatric patients demonstrating a variety alterations in neural oscillations across all major psychiatric diseases, and suggest possible future research directions and treatment approaches based on the fundamental properties of brain rhythms.
High speed and high spatial resolution are at the top of the wish list of every neuroscientist. An important step of progress in this direction has now been made by sampling throughout the brain fMRI signals that temporally surround important physiological patterns.

Activation of type 1 cannabinoid receptors (CB1R) decreases GABA and glutamate release in cortical and subcortical regions, with complex outcomes on cortical network activity. To date there have been few attempts to disentangle the region- and cell-specific mechanisms underlying the effects of cannabinoids on cortical network activity in vivo. Here we addressed this issue by combining in vivo electrophysiological recordings with local and systemic pharmacological manipulations in conditional mutant mice lacking CB1R expression in different neuronal populations. First we report that cannabinoids induce hypersynchronous thalamocortical oscillations while decreasing the amplitude of faster cortical oscillations. Then we demonstrate that CB1R at striatonigral synapses (basal ganglia direct pathway) mediate the thalamocortical hypersynchrony, whereas activation of CB1R expressed in cortical glutamatergic neurons decreases cortical synchrony. Finally we show that activation of CB1 expressed in cortical glutamatergic neurons limits the cannabinoid-induced thalamocortical hypersynchrony. By reporting that CB1R activations in cortical and subcortical regions have contrasting effects on cortical synchrony, our study bridges the gap between cellular and in vivo network effects of cannabinoids. Incidentally, the thalamocortical hypersynchrony we report suggests a potential mechanism to explain the sensory “high” experienced during recreational consumption of marijuana.
This work assigns a prominent role of REM sleep in sleep-related neuronal plasticity by demonstrating how firing rates of hippocampal pyramidal cells and interneurons and their synchrony changes during the sleep cycle.
Spikes contribute to the higher frequency bands of the local field. This work quantified the contribution of spiking to ripple oscillations in the hippocampal CA1 region.
Theta waves are phase shifted by 180o across cell layers, between CA1 and CA3 regions and from the septal to the temporal pole of the hippocampus.
Experimental setup for closed-loop feedback transcranial electrical stimulation (TES). Here, we demonstrate that seizure-triggered, feedback TES can dramatically reduce spike-and-wave episodes in a rodent model of generalized epilepsy. Closed-loop TES can be an effective clinical tool to reduce pathological brain patterns in drug-resistant patients.

When a rat crosses the place field of a hippocampal pyramidal cell, this cell typically fires a series of spikes. Spike phases, measured with respect to theta oscillations of the local field potential, on average decrease as a function of the spatial distance traveled. This relation between phase and position of spikes might be a neural basis for encoding and is called phase precession. The degree of association between the circular phase variable and the linear spatial variable is commonly quantified through, however, a linear-linear correlation coefficient where the circular variable is converted to a linear variable by restricting the phase to an arbitrarily chosen range, which may bias the estimated correlation. Here we introduce a new measure to quantify circular-linear associations. This measure leads to a robust estimate of the slope and phase offset of the regression line, and it provides a correlation coefficient for circular-linear data that is a natural analog of Pearson’s product-moment correlation coefficient for linear-linear data. Using surrogate data, we show that the new method outperforms the standard linear-linear approach with respect to estimates of the regression line and the correlation, and that the new method is less dependent on noise and sample size. We confirm these findings in a large data set of experimental recordings from hippocampal place cells and theta oscillations, and we discuss remaining problems that are relevant for the analysis and interpretation of phase precession. In summary, we provide a new method for the quantification of circular-linear associations.
Neuronal activity in the brain gives rise to transmembrane currents that can be measured in the extracellular medium. We review here the major contributors of the extracellular signal, including the synaptic transmembrane current, Na(+) and Ca(2+) spikes, ionic fluxes through voltage- and ligand-gated channels, and intrinsic membrane oscillations and address the inverse problem of the local field potential. We also hypothesize that high-density LFP recordings can provide covert access to spiking cell assemblies.
High frequency oscillations are ubiquitous in the brain under both physiological conditions and in disease. This review is part of the special issue dedicated to this important topic. The figure shows the emergence and spread of sharp wave ripples from the hippocampus to the entorhinal cortex.
We describe here a diode-probe system that allows real-time and location-specific control of neuronal activity at multiple sites. Manipulation of neuronal activity in arbitrary spatiotemporal patterns is achieved by means of an optoelectronic array, manufactured by attaching multiple diode-fiber assemblies to high-density silicon probes or wire tetrodes, and implanted into the brains of animals that are expressing light-responsive opsins. The capacity of the system to generate synthetic neural activity patterns facilitates multi-site manipulation of neural circuits in a closed-loop manner in the behaving animal.
Because of their ubiquitous nature and strong correlation with the “operational modes” of local circuits, gamma oscillations provide important clues about neuronal population dynamics in health and disease. Here, we review the cellular and synaptic mechanisms underlying gamma oscillations and outline empirical questions and controversial conceptual issues.
Here, we describe and illustrate our methods for recording multiple single neurons and local field potential in behaving rodent; fabrication of microdrives for chronic recordings with silicon probes and our surgical procedures
We introduce a new head-fixed paradigm where all changing stimuli are controlled by the locomotion of the mouse. We demonstrate how division of labor between perisomatic (parvalbumin (PV) expressing) or dendrite-targeting (somatostatin (SOM) expressing) interneurons affect the rate, bursting and timing of pyramidal neurons, using optogenetic methods combined with large-scale silicon probe recordings of unit activity.
This paper is a ‘catalog’ of the various physiological, network and behavioral correlates of firing pattern differences and similarities between CA1 and CA3 pyramidal cells in the rat. The conclusions are based on >3,000 CA1 and >2,000 CA3 pyramidal neurons recorded over the years in our lab in various testing environments.
120 years ago Karoly Schaffer made the important discovery of demonstrating the associational connections between the inferior and superior regions (i.e., CA3 and CA1) of the hippocampus. We review the circumstances of the discovery and the impact it made on hippocampal research.

Neuronal oscillations allow for temporal segmentation of neuronal spikes. Interdependent oscillators can integrate multiple layers of information. We examined phase-phase coupling of theta and gamma oscillators in the CA1 region of rat hippocampus during maze exploration and rapid eye movement sleep. Hippocampal theta waves were asymmetric, and estimation of the spatial position of the animal was improved by identifying the waveform-based phase of spiking, compared to traditional methods used for phase estimation. Using the waveform-based theta phase, three distinct gamma bands were identified: slow gamma(S) (gamma(S); 30-50 Hz), midfrequency gamma(M) (gamma(M); 50-90 Hz), and fast gamma(F) (gamma(F); 90-150 Hz or epsilon band). The amplitude of each sub-band was modulated by the theta phase. In addition, we found reliable phase-phase coupling between theta and both gamma(S) and gamma(M) but not gamma(F) oscillators. We suggest that cross-frequency phase coupling can support multiple time-scale control of neuronal spikes within and across structures.

Neostriatal cholinergic interneurons are believed to be important for reinforcement-mediated learning and response selection by signaling the occurrence and motivational value of behaviorally relevant stimuli through precisely timed multiphasic population responses. An important problem is to understand how these signals regulate the functioning of the neostriatum. Here we describe the synaptic organization of a previously unknown circuit that involves direct nicotinic excitation of several classes of GABAergic interneurons, including neuroptide Y–expressing neurogilaform neurons, and enables cholinergic interneurons to exert rapid inhibitory control of the activity of projection neurons. We also found that, in vivo, the dominant effect of an optogenetically reproduced pause-excitation population response of cholinergic interneurons was powerful and rapid inhibition of the firing of projection neurons that is coincident with synchronous cholinergic activation. These results reveal a previously unknown circuit mechanism that transmits reinforcement-related information of ChAT interneurons in the mouse neostriatal network.
How multiple frequencies in different structures interfere and/or reinforce each other to support behavior is a fascinating question. Here we show how a 4-Hz oscillation in the PFC-VTA axis interact with the hippocampal theta rhythm and how their joint effect can modulate gamma oscillations and unit firing in each of these structures. Such multi level cross-frequency coupling appears an effective mechanism to temporally coordinate neuronal activity in widespread brain areas.
Perhaps the most intensely investigated brain region in the brain is the hippocampal CA1 region. Part of the reasons for this popularity stems from the assumption that this region contains a homogeneous set of pyramidal cells. This work shows that there are at least two sublayers in the CA1 pyramidal layer with distinct biophysical and circuit properties of deep and superficial pyramidal cells. These two circuits can cooperate and segregate information in a brain state-dependent manner. The idea of two sublayers goes back to Schaffer (1882).
Example trace of wide-band traces of ripple (left) and fast gamma (right) oscillation bursts from the CA1 stratum pyramidale (pyr) and radiatum (rad), CA3 pyramidal layer, and dentate granule cell layer (DG S. gran) in a sleeping rat, overlaid on their respective wavelet spectrograms. We examine the cellular, network properties of these respective patterns and show how neocortical-entorhinal inputs can affect their occurrence.

Hippocampal neurons can display reliable and long-lasting sequences of transient firing patterns, even in the absence of changing external stimuli. We suggest that time-keeping is an important function of these sequences, and propose a network mechanism for their generation. We show that sequences of neuronal assemblies recorded from rat hippocampal CA1 pyramidal cells can reliably predict elapsed time (15–20 s) during wheel running with a precision of 0.5 s. In addition, we demonstrate the generation of multiple reliable, long-lasting sequences in a recurrent network model. These sequences are generated in the presence of noisy, unstructured inputs to the network, mimicking stationary sensory input. Identical initial conditions generate similar sequences, whereas different initial conditions give rise to distinct sequences. The key ingredients responsible for sequence generation in the model are threshold-adaptation and a Mexican-hat-like pattern of connectivity among pyramidal cells. This pattern may arise from recurrent systems such as the hippocampal CA3 region or the entorhinal cortex. We hypothesize that mechanisms that evolved for spatial navigation also support tracking of elapsed time in behaviorally relevant contexts.

Axonal arbors of principal neurons form the backbone of neuronal networks in the mammalian cortex. Three-dimensional reconstructions of complete axonal trees are invaluable for quantitative analysis and modeling. However, digital data are still sparse due to labor intensity of reconstructing these complex structures. We augmented conventional tracing techniques with computational approaches to reconstruct fully labeled axonal morphologies. We digitized the axons of three rat hippocampal pyramidal cells intracellularly filled in vivo from different CA3 sub-regions: two from areas CA3b and CA3c, respectively, toward the septal pole, and one from the posterior/ventral area (CA3pv) near the temporal pole. The reconstruction system was validated by comparing the morphology of the CA3c neuron with that traced from the same cell by a different operator on a standard commercial setup. Morphometric analysis revealed substantial differences among neurons. Total length ranged from 200 (CA3b) to 500 mm (CA3c), and axonal branching complexity peaked between 1 (CA3b and CA3pv) and 2 mm (CA3c) of Euclidean distance from the soma. Length distribution was analyzed among sub-regions (CA3a,b,c and CA1a,b,c), cytoarchitectonic layers, and longitudinal extent within a three-dimensional template of the rat hippocampus. The CA3b axon extended thrice more collaterals within CA3 than into CA1. On the contrary, the CA3c projection was double into CA1 than within CA3. Moreover, the CA3b axon extension was equal between strata oriens and radiatum, while the CA3c axon displayed an oriens/radiatum ratio of 1:6. The axonal distribution of the CA3pv neuron was intermediate between those of the CA3b and CA3c neurons both relative to sub-regions and layers, with uniform collateral presence across CA3/CA1 and moderate preponderance of radiatum over oriens. In contrast with the dramatic sub-region and layer differences, the axon longitudinal spread around the soma was similar for the three neurons. To fully characterize the axonal diversity of CA3 principal neurons will require higher-throughput reconstruction systems beyond the threefold speed-up of the method adopted here.
Although glucose is the primary energetic substrate of the brain, it has been debated whether neurons directly metabolize glucose, or alternatively, glucose is broken down to lactate by astrocytes, which feed the neurons by lactate. We examined these hypotheses by examining the incorporation of a nonmetabolizable fluorescent glucose analog (green in the figure; astrocytes red), using 2-photon laser scanning microscopy in the rat S1 cortex in vivo. Whisker stimulation lead to a quick incorporation of the glucose analog in astrocytes but much less so in neurons. These results provide direct support for the glia-neuron “lactate-shuttle” hypothesis of Magistretti and Pellerin put forward 15 years ago. Progress takes time!
While all neuroscientists agree that neurons in the brain come together temporally to form coalitions (‘cell assemblies’), there is neither an accepted definition of such hypothetical neuronal assemblies nor a strategy how to identify them. This review suggests that assemblies make sense only from the viewpoint of a goal (referred to as an ‘observer or reader’ mechanism) and discusses potential experimental paradigms for a disciplined way to study them.
Transcranial electrical stimulation (TES) can produce diffuse currents in the brain and lead to stimulus-locked firing in cortical neurons. The figure shows various configurations of the extracranial electrodes in rats, and the impact of sinusoid ~1 Hz TES on extracellular and intracellular activities. In addition as an experimental tool, TES is an ideal method for therapeutic control of brain activity.
The entorhinal cortex is an important generator theta oscillations and gives rise to interesting dynamics, such as the ‘grid’ cells of the Mosers. This work provides critical anatomical data about the in vivo connectivity of layer 2, 3 and 5 neurons in the entorhinal cortex of the rat and shows that stellate cells are critical components of theta generation. NeuroLucida data of the reconstructed neurons are available upon request.
This paper describes novel methods for delivering light to a very small volume of brain tissue using the combination of etched optic fibers with large scale recording of neurons by silicon probes in behaving rats and mice. These methods open new possibilities to perturb the local circuit in a controlled manner, activating or silencing only those neurons whose activity is constantly being monitored as well. This work was performed at my ‘satellite’ lab at Janelia Farm, in collaboration with Jeff Magee and his excellent people.
Spikes of nearly all hippocampal place cells and episode cells oscillate faster than the oscillation frequency of the simultaneously recorded population (also reflected by the LFP theta; as shown by this figure). Our model shows why: the temporal delays between place cells (according to a ‘compression’ rule; i.e., distance representations are proportional to the within-theta spike time delays) shift the population frequency lower. The model accounts for all known major features of hippocampal place cells.
The cooperative action of neurons and glia generates electrical fields, but their effect on individual neurons via ephaptic interactions is mostly unknown. Here, we analyze the impact of spatially inhomogeneous electric fields on the membrane potential, the induced membrane field, and the induced current source density of one-dimensional cables as well as morphologically realistic neurons and discuss how the features of the extracellular field affect these quantities. We show through simulations that endogenous fields, associated with hippocampal theta and sharp waves, can greatly affect spike timing. These findings imply that local electric fields, generated by the cooperative action of brain cells, can influence the timing of neural activity.
While the hippocampus is a giant single module its inputs, outputs, local wiring and intrinsic properties of neurons differ along the septo-temporal axis. Recording from neurons in dorsal and ventralmost part of the hippocampus, we find numerous quantitative and qualitative differences between these populations. Theta rhythmicity was less pronounced in the ventral part, yet theta oscillations phase modulated gamma power at all locations. Thus, theta may bind together the results local computations into a more global pattern.
How multiple frequencies in different structures interfere and/or reinforce each other to support behavior is a fascinating question. Here we show how a 4-Hz oscillation in the PFC-VTA axis interact with the hippocampal theta rhythm and how their joint effect can modulate gamma oscillations and unit firing in each of these structures. Such multi level cross-frequency coupling appears an effective mechanism to temporally coordinate neuronal activity in widespread brain areas.

During the crossing of the place field of a pyramidal cell in the rat hippocampus, the firing phase of the cell decreases with respect to the local theta rhythm. This phase precession is usually studied on the basis of data in which many place field traversals are pooled together. Here we study properties of phase precession in single trials. We found that single-trial and pooled-trial phase precession were different with respect to phase-position correlation, phase-time correlation, and phase range. Whereas pooled-trial phase precession may span 360°, the most frequent single-trial phase range was only ∼180°. In pooled trials, the correlation between phase and position (r = −0.58) was stronger than the correlation between phase and time (r = −0.27), whereas in single trials these correlations (r = −0.61 for both) were not significantly different. Next, we demonstrated that phase precession exhibited a large trial-to-trial variability. Overall, only a small fraction of the trial-to-trial variability in measures of phase precession (e.g., slope or offset) could be explained by other single-trial properties (such as running speed or firing rate), whereas the larger part of the variability remains to be explained. Finally, we found that surrogate single trials, created by randomly drawing spikes from the pooled data, are not equivalent to experimental single trials: pooling over trials therefore changes basic measures of phase precession. These findings indicate that single trials may be better suited for encoding temporally structured events than is suggested by the pooled data.
How multiple frequencies in different structures interfere and/or reinforce each other to support behavior is a fascinating question. Here we show how a 4-Hz oscillation in the PFC-VTA axis interact with the hippocampal theta rhythm and how their joint effect can modulate gamma oscillations and unit firing in each of these structures. Such multi level cross-frequency coupling appears an effective mechanism to temporally coordinate neuronal activity in widespread brain areas.

It’s been known for more than a century that sleep somehow is important for learning and memory. Sigmund Freud further suspected that what we learned during the day was ‘rehearsed’ by the brain during dreaming, allowing memories to form. But while much recent research has focused on the correlative links between the hippocampus and memory consolidation during sleep, the specific processes that cause long-term memories to form has not been identified. Because sharp wave ripples have been implicated as the key mechanism for transferring transient hippocampal traces for long-term storage in the neocortex (Buzsaki, 1989), we decided to kill all ripples during post-learning sleep. While specific and selective elimination hippocampal ripples did not affect the structure of sleep, it prevented the rats from efficiently learning a hippocampus-dependent maze task. Identification of a specific brain pattern responsible for strengthening learned information could facilitate applied research for more effective treatment of memory disorders. (This image was designed by MZ but NN chose an ugly cover instead of this).
The macroscopic theta oscillation is a result of multiple cooperating theta dipoles. Each layer in each hippocampal region represents a theta current source and the cooperation of these dipoles varies as a function of behavior. Therefore, for the demonstration of a relationship between theta oscillations and behavior/neuronal activity, precise regional verification of the current sources is required.

The hippocampus provides a spatial map of the environment. Changes in the environment alter the firing patterns of hippocampal neurons, but are presumably constrained by elements of the network dynamics. We compared the neural activity in CA1 and CA3 regions of the hippocampus in rats running for water reward on a linear track, before and after the track length was shortened. A fraction of cells lost their place fields and new sets of cells with fields emerged, indicating distinct representation of the two tracks. Cells active in both environments shifted their place fields in a location-dependent manner, most notably at the beginning and the end of the track. Furthermore, peak firing rates and place-field sizes decreased, whereas place-field overlap and coactivity increased. Power in the theta-frequency band of the local field potentials also decreased in both CA1 and CA3, along with the coherence between the two structures. In contrast, the theta-scale (0-150 ms) time lags between cell pairs, representing distances on the tracks, were conserved, and the activity of the inhibitory neuron population was maintained across environments. We interpret these observations as reflecting the freedoms and constraints of the hippocampal network dynamics. The freedoms permit the necessary flexibility for the network to distinctly represent unique patterns, whereas the dynamics constrain the speed at which activity propagates between the cell assemblies representing the patterns.
The picture illustrates oscillatory ocean waves, which give rise to surges of local water splashes on the peak of the wave, akin to how theta oscillation biases the occurrence of gamma oscillation splashes throughout the cortex at the peak of hippocampal theta oscillation. We demonstrate how hippocampal theta oscillations modulate neocortical unit activity and gamma rhythms in rats during exploration and REM sleep. This study suggests a mechanism by which hippocampal theta creates the temporal windows of opportunity within which the multiple local computation in local neocortical circuits (gamma oscillations) can be effectively transferred to the hippocampus (photo: Pascale Quilichini).

Technological advances in neural interfaces are providing increasingly more powerful “toolkits” of designs, materials, components, and integrated devices for establishing high-fidelity chronic neural interfaces. For a broad class of neuroscience studies, the primary requirements of these interfaces include recording and/or stimulating from a number of discretely sampled volumes at requisite spatial resolutions for specific periods of time. Translational and clinical applications present additional requirements for safety, usability, reliability, patient acceptance, and cost effectiveness. Innovative solutions result from the constructive tension between ever-increasing application requirements and incorporation of technological advances into usable devices. The purpose of this minireview is to present snapshots of the current state-of-the-art in chronic, microscale neural interfaces by highlighting several leading neuroscience applications and discussing their implications for next-generation interface devices.
A long-standing conjecture in neuroscience is that aspects of cognition depend on the brain’s ability to self-generate sequential neuronal activity. We found that reliably and continually changing cell assemblies in the rat hippocampus appeared not only during spatial navigation but also in the absence of changing environmental or body-derived inputs. During the delay period of a memory task, each moment in time was characterized by the activity of a particular assembly of neurons. Identical initial conditions triggered a similar assembly sequence, whereas different conditions gave rise to different sequences, thereby predicting behavioral choices, including errors. Such sequences were not formed in control (nonmemory) tasks. We hypothesize that neuronal representations, evolved for encoding distance in spatial navigation, also support episodic recall and the planning of action sequences.
REM sleep is the most mysterious state of the brain. The brain loses its control over muscles of the body and the environmental inputs are ignored. During this self-isolated steady state short bursts of motor outputs occur. We report here that these phasic bursts of REM are characterized by enhanced throroughputs in hippocampal networks. One can only speculate that the compressed neuronal contents of the hippocampus during the phasic events may be responsible for generating dream content of REM. (Image: Flaming-June by FrederickLeighton, Ponce Museum of Art).
Neuroscience produces a vast amount of data from an enormous diversity of neurons. A neuronal classification system is essential to organize such data and the knowledge that is derived from them. Classification depends on the unequivocal identification of the features that distinguish one type of neuron from another. The problems inherent in this are particularly acute when studying cortical interneurons. To tackle this, we convened a representative group of researchers to agree on a set of terms to describe the anatomical, physiological and molecular features of GABAergic interneurons of the cerebral cortex. The resulting terminology might provide a stepping stone towards a future classification of these complex and heterogeneous cells. Consistent adoption will be important for the success of such an initiative, and we also encourage the active involvement of the broader scientific community in the dynamic evolution of this project.
A neural coding scheme formed by the combined function of gamma and theta oscillations. Lisman J, Buzsáki G. Schizophr Bull 2008 Sep; 34 (5):974-80

In rodent hippocampus, neuronal activity is organized by a 6–10 Hz theta oscillation. The spike timing of hippocampal pyramidal cells with respect to the theta rhythm correlates with an animal’s position in space. This correlation has been suggested to indicate an explicit temporal code for position. Alternatively, it may be interpreted as a byproduct of theta-dependent dynamics of spatial information flow in hippocampus. Here we show that place cell activity on different phases of theta reflects positions shifted into the future or past along the animal’s trajectory in a two-dimensional environment. The phases encoding future and past positions are consistent across recorded CA1 place cells, indicating a coherent representation at the network level. Consistent theta-dependent time offsets are not simply a consequence of phase-position correlation (phase precession), because they are no longer seen after data randomization that preserves the phase-position relationship. The scale of these time offsets, 100–300 ms, is similar to the latencies of hippocampal activity after sensory input and before motor output, suggesting that offset activity may maintain coherent brain activity in the face of information processing delays.
Representation of the world and internal states of the brain are usually thought to be expressed in the firing patterns of neurons. However, short-term synaptic dynamics are equally effective in temporally storing information but experimental exploration of this mechanism in the behaving animal has not been possible until recently. High density recordings from local circuits with silicon probes allows access to this important coding mechanism. This paper demonstrates how synaptic weights vary dynamically according to task demands.
Both the thalamocortical and limbic systems generate a variety of brain state-dependent rhythms but the relationship between the oscillatory families is not well understood. Transfer of information across structures can be controlled by the offset oscillations. We suggest that slow oscillation of the neocortex, which was discovered by Mircea Steriade, temporally coordinates the self-organized oscillations in the neocortex, entorhinal cortex, subiculum and hippocampus. Transient coupling between rhythms can guide bidirectional information transfer among these structures and might serve to consolidate memory traces.
Functional MRI is the most important window for probing the human brain. However, the neuronal events correlated with the BOLD signal (‘activation maps’) are not well understood. Here we discuss why understanding the role of inhibition in brain activity is so critical for the interpretation of such a ‘mean field’ signal. This review is a synthesis of nearly 3 years of intense discussion among the three of us (GB, KK, MR) with different expertise and perspectives on the brain. I enjoyed our interactions a lot and would like to thank Kai and Marc for being so patient and emphatic teachers. Image: Interneurons are fueled mainly by mitochondrium-dependent (arrows) oxidate phosphorilation.
Abstract depiction of forward and reverse place-cell sequences. Each column represents a window in time, with subthreshold and suprathreshold place-cell activity. Sequences sampled in the middle are played out in full, in forward at the beginning, and in reverse at the end.
The function of hilar mossy cells has remained a mystery. The collection of papers in this volume attempt to address their role.
Gamma power in the CA1 str. radiatum and gamma coherence between CA3 and CA1 regions is consistently highest in the central (choice) arm of a T maze. These findings suggest that recall preferentially engages the CA3-CA1 associational system.
Single CA3 neurons innervate from one half to two thirds of the hippocampus and contact targets in a relatively random manner. The implication is that the CA3-CA1 system represent a single large network module (with the possible separation of the ventral tip).
In this collaborative research with Peter Somogyi’s group, we identified several types of long-range GABAergic interneurons. In a restrictive sense, the term “interneuron” does not really here apply since these cells have as extensive axon arbors as those of the pyramidal cells, spanning regions and various structures. These long-range interneurons may serve as “short cuts” for converting the locally organized interneurons into a “small world network” (Buzsaki et al., TINS 2004). The large caliber and strong myelination of the projection axon of the long-range interneurons, relative to those of pyramidal cells (Figure) allow for an especially fast communication between neuronal assemblies.
Hippocampal place cells are speed-controlled oscillators. They oscillate faster than the “baseline” field theta and produce an interference pattern (“phase precession” – O’Keefe and Recce, 1993).
Subjective awareness may depend on neural networks in the brain supporting complex wiring schemes and dynamic patterns of activity.
Perhaps nowhere is the truism ‘structure defines function’ more appropriate than for the brain. The architecture of different brain regions determines the kinds of computations that can be carried out, and may dictate whether a particular region can support subjective awareness. Also, the degree of architectural complexity may determine susceptibility to neurological and psychiatric diseases — complex architectural schemes being more prone to disruption than simpler ones. Understanding how such structure–function relationships govern brain operations requires a systems-level approach that explores how local computation relates to global patterns of neural activity.
How can drug discovery for psychiatric disorders be improved? Agid Y, Buzsáki G, Diamond DM, Frackowiak R, Giedd J, Girault JA, Grace A, Lambert JJ, Manji H, Mayberg H, Popoli M, Prochiantz A, Richter-Levin G, Somogyi P, Spedding M, Svenningsson P, Weinberger D. Nat Rev Drug Discov 2007 Mar; 6 (3):189-201
Cortical control of zona incerta. Barthó P, Slézia A, Varga V, Bokor H, Pinault D, Buzsáki G, Acsády L. J Neurosci 2007 Feb 14; 27 (7):1670-81
Large areas of the neocortex and paleocortex reboot their activity several thousand times every night. Each delta wave (or DOWN state) is followed by an organized sequence neuronal recruitment rather than a random pattern of activity. The UP-state initiator neurons may gain their critical role from either their intrinsic properties or by their stronger functional connectivity in the network(s) they are embedded., and, potentially, may reflect experience-dependent effects. The temporal precision of the recruited neurons along the sequence decreases according to a power law.
Large areas of the neocortex and paleocortex reboot their activity several thousand times every night. Each delta wave (or DOWN state) is followed by a period of sustained activity at a frequency of 0.5-1.5 Hz (i.e., the slow oscillation of Steriade et al., 1993). Granule cells of the dentate gyrus are also ‘enslaved’ to the slow oscillation. Although the firing patterns of CA3 and CA1 region neurons are also biased temporally, the CA3 region can give rise to self-generated patterns also during the DOWN state. Thus, the dentate-CA3 interface is a major functional gate.
The active ingredient in marijuana (tetrahydrocannabinoid or THC) and a synthetic cannabinoid agonist interfere with synchronized activity between neurons in the hippocampus of rats. We recorded from multiple neurons in the hippocampus of rats. Normally neurons in this region form groups that fire action potentials in the time windows of gamma and theta oscillations. But when CB1 agonist was administered, this synchrony was disrupted. The drug did not change the total number of action potentials produced, just their tendency to occur synchronously in these time windows. Animals with less synchronized neural activity under the drug performed less well in a hippocampus-dependent test of memory, suggesting that synchronized neural firing is essential for normal hippocampal function.
Morphology and synaptic input of substance P receptor-immunoreactive interneurons in control and epileptic human hippocampus.
Tóth K, Wittner L, Urbán Z, Doyle WK, Buzsáki G, Shigemoto R, Freund TF, Maglóczky Z. Neuroscience 2007 Jan 19; 144 (2):495-508
Hippocampal CA3 pyramidal cells selectively innervate aspiny interneurons. Wittner L, Henze DA, Záborszky L, Buzsáki G. Eur J Neurosci 2006 Sep; 24 (5):1286-98
Populations of hippocampal inhibitory neurons express different levels of cytochrome c. Gulyás AI, Buzsáki G, Freund TF, Hirase H. Eur J Neurosci 2006 May; 23 (10):2581-94
Among the most remarkable features of a memory episode is the sequential ordering of composite events and the spatial-temporal relationships that bind them together into a unique episode. Similar to episodic learning of serial events, sequential activation of hippocampal place cells during rat movement on a track is believed to produce a representation that binds past, present, and future locations into a spatial episode. Dragoi and Buzsaki show that compressed spatial place cell sequences are represented for several theta cycles by the temporally coordinated activity of hippocampal cell assemblies, with the activity of CA3 preceding the activity of CA1 assemblies by one-half of a theta cycle. The results suggest a role for CA3 in binding CA1 temporal place cell sequences into single episodes. The context and implications of this study and related recent work on hippocampal place cell assemblies are discussed in a Minireview by Suzuki.
Klusters, NeuroScope, NDManager: a free software suite for neurophysiological data processing and visualization. Hazan L, Zugaro M, Buzsáki G. J Neurosci Methods 2006 Sep 15; 155 (2):207-16 [PDF]
On the origin of the extracellular action potential waveform: A modeling study. Gold C, Henze DA, Koch C, Buzsáki G. J Neurophysiol 2006 May; 95 (5):3113-28
Synaptic plasticity and self-organization in the hippocampus. Buzsáki G, Chrobak JJ. Nat Neurosci 2005 Nov; 8 (11):1418-20
Representation of information in the hippocampus occurs at multiple spatio-temoporal scale, symbolized here by the fractal nature of the hippocampus. Superimposed are one-second segments of field potentials from 96-sites recorded simultaneously in the dorsal hippocampus. The behavioral-physiological functions of theta oscillations have been debated over the past 70 years and the current views are summarized in this issue.
Neuroscience: neurons and navigation. Buzsáki G, Nature 2005 Aug 11; 436 (7052):781-2
Band-tunable and multiplexed integrated circuits for simultaneous recording and stimulation with microelectrode arrays.
Olsson RH 3rd, Buhl DL, Sirota AM, Buzsaki G, Wise KD. IEEE Trans Biomed Eng 2005 Jul; 52 (7):1303-11 [PDF]
Neuroscience. Similar is different in hippocampal networks. Buzsáki G, Science 2005 Jul 22; 309 (5734):568-9
Developmental emergence of hippocampal fast-field “ripple” oscillations in the behaving rat pups. Buhl DL, Buzsáki G. Neuroscience 2005; 134 (4):1423-30 [PDF]
Performance of real networks can be simulated by a very large number computational models with various architectures, algorithms and expected differential responses to perturbations. The real challenge then is to identify the right model. Various computational, single cell and slice models have been offered for “phase precession”, a potential phase-coding method used by the hippocampus to identify spatio-temporal relationship of sequential events. In order to examine the contribution of the hippocampal networks in this process, we perturbed progression of phase-precession of single place neurons but “switching off” the hippocampus for ~ 100-200 msec and resetting the phase of global theta oscillation. Despite these manipulations, hippocampal activity reorganized instantaneously and phase procession continued. These findings suggest that the hippocampal information is updated by every theta cycle. A “biologically relevant” model of phase-coding should behave similarly.

How do you know where your nose is? In a more general context, you may ask: how does the sensory representation of the cortex acquire its real-world 3-dimensional representation of the world? Our short answer is that the 3-dimensional geometric layout of the skeletal muscle system is responsible for training the somatosensory system during early development. This stage coincides with the first organized cortical pattern in the form of a spindle-shaped rhythm, fine turning of local connectivity and the emergence of long-range cortico-cortical and cortico-spinal axons. Stochastic, isolated muscle twitches, limb movements and whole body jerks, triggered by autonomous spinal cord circuits in the later stages of pregnancy (human) and first week of life (rat), serve as a supervised algorithm that decreases the infinite possibilities of sensory input combinations to the minimum that will be used in later life. The movement – reafferentation – prolonged cortical activation sequence is among the finest examples of self-organization in nature.

The importance of long-term synaptic plasticity as a cellular substrate for learning and memory is well established. By contrast, little is known about how learning and memory are regulated by voltage-gated ion channels that integrate this synaptic information. We investigated this question using mice with general or forebrain-restricted knockout of the HCN1 gene, which we find encodes a major component of the hyperpolarization-activated inward current (Ih) and is an important determinant of dendritic integration in hippocampal CA1 pyramidal cells. Surprisingly, deletion of HCN1 from forebrain neurons enhances hippocampal-dependent spatial learning and memory. Deletion of HCN1 augments the power of theta oscillation in the CA1 region and enhances long-term potentiation (LTP) at the distal cortical inputs to CA1 neurons, where HCN1 is most strongly expressed, but has little effect on synaptic integration or LTP at the more proximal Schaffer collateral inputs. We suggest that HCN1 channels constrain spatial learning and memory by regulating dendritic integration of distal synaptic inputs.
The Local hemodynamics of the cerebral cortex is the basis of modern functional imaging techniques, such as fMRI and PET. Despite the importance of local regulation of the blood flow, capillary level quantification of cerebral blood flow has been limited by the spatial resolution of functional imaging techniques and the depth penetration of conventional optical microscopy. He we use two-photon laser scanning microscopic imaging technique to monitor basal capillary flux in mice as a function of neuronal activity. Our results show that local hyper-synchronized neural activity is associated with increased capillary perfusion in a volume that is significantly smaller than the currently available resolution of the fMRI signal.
If you have seen Luis Bravo’s extravaganza “Forever Tango” you can picture the qualitative essence of synchrony through rhythm: coupling through time by weak links. Instead of brute force, subtle facial expressions, harmonic body movement, light touch and other invisible magic link the partners swing in perfect unison. In the brain, synchrony can also be brought about by force, e.g. by strong excitation through glutamate receptors. Alternatively, it can emerge by phase coupling of oscillatory ensembles through weak links, such as the sparse long-range connections between cortical areas. We speculate that oscillation-based synchrony is an essential part of the brain’s design that serves numerous useful functions.
Recording from statistically representative samples of identified neurons from several local areas while minimally interfering with the brain activity is a major goal in systems neuroscience. Many other methods are available for studying the brain but in the end all these indirect observations should be translated back into a common currency – the format of neuronal spike trains – to understand the brain’s control of behavior. Wire “tetrodes” and silicon probes can ‘hear’ pyramial cells as far away as 140um lateral in the cell body. A cylinder with a radius of 140 um contains ~1,000 neurons in the rat cortex, which is the number of theoretically recordable cells by a single electrode. High-density silicon probes and novel mathematical methods in the future may allow us to record all neurons in this volume.
Large and long lasting cytosolic calcium surges in astrocytes have been described in cultured cells and acute slice preparations.The mechanisms that give rise to these calcium events have been extensively studied in vitro. However, their existence and functions in the intact brain are unknown. We have topically applied fluo-4 AM on the cerebral cortex of anesthetized rats and imaged cytosolic calcium fluctuation in astrocyte populations of superficial cortical layers in vivo, using 2-photon laser scanning microscopy. Spontaneous [Ca2+]i events in individual astrocytes were similar to those observed in vitro. Coordination of [Ca2+]i events among astrocytes was indicated by the broad cross-correlograms. Increased neuronal discharge was associated with increased astrocytic [Ca2+]i activity in individual cells and a robust coordination of [Ca2+]i signals in neighboring astrocytes. These findings provide indicate potential neuron-glia communication in the intact brain.
Separation of principal cells and GABAergic interneurons is of utmost importance for the interpretation of neuronal interactions in the cortex. The cover shows high-density recording of neurons in layer 5 of the somatosensory cortex, using 8-shank, 64-site two-dimensional silicon probes. From their short-term synaptic interaction, units could be identified as excitatory or inhibitory. Large-scale recordings of neuronal activity, determination of their physical location in the cortex and their classification into pyramidal and interneuron classes provide the necessary tools for local circuit analysis. From: Bartho P, Hirase H, Monconduit L, Zugaro M, Harris KD, and Buzsaki G. Characterization of neocortical principal cells and interneurons by network interactions and extracellular features.
The The performance of the brain is constrained by wiring length and maintenance costs. The apparently inverse relationship between number of neurons in the various interneuron classes and the spatial extent of their axon tree suggests a mathematically definable organization, reminiscent of “small-world” or scale-free networks observed in other complex systems. The wiring economy-based classification of cortical inhibitory interneurons is supported by the distinct physiological patterns of class members in the intact brain. We hypothesize that the complex wiring of diverse interneuron classes represents an economic solution for supporting global synchrony and oscillations at multiple time scales with minimum axon length.
Image: Detail from Mark Lombardi’s now famous pencil drawing: Oliver North, Lake Resources of Panama, and the Iran-Contra Operation, 1984-86). Lombardi’s artistic graphs capture the essence of small-world networks, a feature utilized by brain connectivity.
Pericytes in the central nervous system (CNS) are hypothesized to be involved in important circulatory functions, including local blood flow regulation, angiogenesis, immune reaction, and regulation of blood-brain barrier. Despite these putative functions, functional correlates of pericytes in vivo are scarce. We have labeled CNS pericytes using the dextran-conjugated fluorescent calcium indicator Calcium Green I and imaged them in somatosensory cortex of the mouse in vivo. Intracellular calcium concentration in pericytes showed spontaneous surges lasting for several seconds. Furthermore, population bursts of neuronal activity were associated with increased Ca2+ signal in a portion of the pericytes. Selective in vivo labeling of pericytes with functional markers may help reveal their physiological function in neuronal activity-associated regulation of local cerebral blood flow.
In the brain, information is encoded by the firing patterns of neuronal ensembles and the strength of synaptic connections between individual neurons. We report here that representation of the environment by “place” cells is altered by changing synaptic weights within hippocampal networks. Long-term potentiation (LTP) of intrinsic hippocampal pathways abolished existing place fields, created new place fields, and rearranged the temporal relationship within the affected population. The effect of LTP on neuron discharge was rate and context dependent. The LTP-induced “remapping” occurred without affecting the global firing rate of the network. The findings support the view that learned place representation can be accomplished by LTP-like synaptic plasticity within intrahippocampal networks.
A short segment of wide band recording from layer V of the somatosensory cortex of the anesthetized rat during down-up-down state transition. 64-site recording with an 8-shank silicon probe. Recordings from separate shanks (8 recording sites each) are distinguished by alternating colors. Background shows detail of the preamplifier circuit integrated in the recording probe.
Large-scale recording of neuronal activity in the hippocampus revealed assembly organization. Prediction of the timing of pyramidal cells spikes is improved using the spike times of peer neurons, over prediction from the animal’s trajectory in space, or a spatially-dependent theta phase modulation. Coordinated variability of spike timing in cell assemblies is assumed to represent a brain-derived (cognitive) process.
Brain-state- and cell-type-specific firing of hippocampal interneurons in vivo. Klausberger T, Magill PJ, Márton LF, Roberts JD, Cobden PM, Buzsáki G, Somogyi P. Nature 2003 Feb 20; 421 (6925):844-8 [PDF]
Communication between neocortex and hippocampus during sleep in rodents. Sirota A, Csicsvari J, Buhl D, Buzsáki G. Proc Natl Acad Sci U S A 2003 Feb 18; 100 (4):2065-9 [PDF]
Selective impairment of hippocampal gamma oscillations in connexin-36 knock-out mouse in vivo. Buhl DL, Harris KD, Hormuzdi SG, Monyer H, Buzsáki G. J Neurosci 2003 Feb 1; 23 (3):1013-8 [PDF]
The cover shows data obtained from two-dimensional recording of gamma oscillation in various hippocampal regions of the behaving rat. The six-shank silicon probe, with 16 recording sites each, is placed in the CA1-CA3 and dentate regions. The derived voltage traces are converted into currents to illustrate large amplitude and coherent gamma oscillatory activity in the CA3-CA1 pyramidal layer. A coronal section of the hippocampus stained with the Gallyas silver method (dark field photograph) is shown in the background.

The physiological roles of neuronal gap junctions in the intact brain are not known. The recent generation of the connexin-36 knockout (Cx36 KO) mouse has offered a unique opportunity to examine this problem. Recent in vitro recordings in Cx36 KO mice suggested that Cx36 gap junction contributes to various oscillatory patterns in the theta (~5-10 Hz) and gamma (~30-80 Hz) frequency ranges, and affects certain aspects of high frequencies (>100 Hz) patterns. However, the relevance of these pharmacologically-induced patterns to the intact brain is not known. We recorded field potentials and unit activity in the CA1 stratum pyramidale of the hippocampus in behaving of wild type (WT) and Cx36 KO mice. Fast-field ripple oscillations (140-200 Hz) were present in both WT and KO mice and did not differ significantly in power, intraepisode frequency or probability of occurrence. Thus, fast-field oscillations may either not require electrical synapses or may be mediated by a hitherto unknown class of gap junctions. Theta oscillations, recorded during either wheel running or rapid eye movement sleep, were not different either. However, the power in the gamma frequency band and the magnitude of theta-phase modulation of gamma power were significantly decreased in KO mice compared to WT controls during wheel running. This suggests that Cx36 interneuronal gap junctions selectively contribute to gamma oscillations.
Homeostatic maintenance of neuronal excitability by burst discharges in vivo. Buzsáki G, Csicsvari J, Dragoi G, Harris K, Henze D, Hirase H. Cereb Cortex 2002 Sep; 12 (9):893-9 [PDF]
Single granule cells reliably discharge targets in the hippocampal CA3 network in vivo. Henze DA, Wittner L, Buzsáki G. Nat Neurosci 2002 Aug; 5 (8):790-5 [PDF]
Spike train dynamics predicts theta-related phase precession in hippocampal pyramidal cells. Harris KD, Henze DA, Hirase H, Leinekugel X, Dragoi G, Czurkó A, Buzsáki G. Nature 2002 Jun 13; 417 (6890):738-41 [PDF]
Correlated bursts of activity in the neonatal hippocampus in vivo. Leinekugel X, Khazipov R, Cannon R, Hirase H, Ben-Ari Y, Buzsáki G. Science 2002 Jun 14; 296 (5575):2049-52 [PDF]
Theta oscillations in the hippocampus. Buzsáki G, Neuron 2002 Jan 31; 33 (3):325-40 [PDF]
Hippocampal pyramidal cell-interneuron spike transmission is frequency dependent and responsible for place modulation of interneuron discharge. Marshall L, Henze DA, Hirase H, Leinekugel X, Dragoi G, Buzsáki G. J Neurosci 2002 Jan 15; 22 (2):RC197
Hippocampal GABAergic interneurons: a physiological perspective. Buzsáki G, Neurochem Res 2001 Sep; 26 (8-9):899-905
Temporal interaction between single spikes and complex spike bursts in hippocampal pyramidal cells. Harris KD, Hirase H, Leinekugel X, Henze DA, Buzsáki G. Neuron 2001 Oct 11; 32 (1):141-9 [PDF]
Electrical wiring of the oscillating brain. Buzsáki G, Neuron 2001 Aug 16; 31 (3):342-4
Action potential threshold of hippocampal pyramidal cells in vivo is increased by recent spiking activity. Henze DA, Buzsáki G. Neuroscience 2001; 105 (1):121-30 [PDF]
Firing rates of hippocampal neurons are preserved during subsequent sleep episodes and modified by novel awake experience. Hirase H, Leinekugel X, Czurkó A, Csicsvari J, Buzsáki G. Proc Natl Acad Sci U S A 2001 Jul 31; 98 (16):9386-90
Behavior-dependent states of the hippocampal network affect functional clustering of neurons. Hirase H, Leinekugel X, Csicsvari J, Czurkó A, Buzsáki G. J Neurosci 2001 May 15; 21 (10):RC145
The apical shaft of CA1 pyramidal cells is under GABAergic interneuronal control. Papp E, Leinekugel X, Henze DA, Lee J, Buzsáki G. Neuroscience 2001; 102 (4):715-21 [PDF]
The application of printed circuit board technology for fabrication of multi-channel micro-drives. Szabó I, Czurkó A, Csicsvari J, Hirase H, Leinekugel X, Buzsáki G. J Neurosci Methods 2001 Jan 30; 105 (1):105-10 [PDF]
Ensemble patterns of hippocampal CA3-CA1 neurons during sharp wave-associated population events. Csicsvari J, Hirase H, Mamiya A, Buzsáki G. Neuron 2000 Nov; 28 (2):585-94
Unusual target selectivity of perisomatic inhibitory cells in the hilar region of the rat hippocampus. Acsády L, Katona I, Martínez-Guijarro FJ, Buzsáki G, Freund TF. J Neurosci 2000 Sep 15; 20 (18):6907-19 [PDF]
Physiological patterns in the hippocampo-entorhinal cortex system. Chrobak JJ, Lörincz A, Buzsáki G. Hippocampus 2000; 10 (4):457-65 [PDF]
Two-phase computational model training long-term memories in the entorhinal-hippocampal region. Lörincz A, Buzsáki G. Ann N Y Acad Sci 2000 Jun; 911 83-111
Accuracy of tetrode spike separation as determined by simultaneous intracellular and extracellular measurements. Harris KD, Henze DA, Csicsvari J, Hirase H, Buzsáki G. J Neurophysiol 2000 Jul; 84 (1):401-14 [PDF]
Intracellular features predicted by extracellular recordings in the hippocampus in vivo. Henze DA, Borhegyi Z, Csicsvari J, Mamiya A, Harris KD, Buzsáki G. J Neurophysiol 2000 Jul; 84 (1):390-400[PDF]
Multiple site silicon-based probes for chronic recordings in freely moving rats: implantation, recording and histological verification.
Bragin A, Hetke J, Wilson CL, Anderson DJ, Engel J Jr, Buzsáki G. J Neurosci Methods 2000 May 15; 98 (1):77-82[PDF]
Firing rate and theta-phase coding by hippocampal pyramidal neurons during ‘space clamping’. Hirase H, Czurko A, Csicsvari J, Buzsaki G. Eur J Neurosci 1999 Dec; 11(12):4373-80
Ultra-slow oscillation (0.025 Hz) triggers hippocampal afterdischarges in Wistar rats. Penttonen M, Nurminen N, Miettinen R, Sirvio J, Henze DA, Csicsvari J, Buzsaki G. Neuroscience 1999; 94(3):735-43
Hebbian modification of a hippocampal population pattern in the rat. King C, Henze DA, Leinekugel X, Buzsaki G. J Physiol 1999 Nov; 521 Pt 1():159-67
Replay and time compression of recurring spike sequences in the hippocampus. Nadasdy Z, Hirase H, Czurko A, Csicsvari J, Buzsaki G. J Neurosci 1999 Nov; 19(21):9497-507
Fast network oscillations in the hippocampal CA1 region of the behaving rat. Csicsvari J, Hirase H, Czurko A, Mamiya A, Buzsaki G. J Neurosci 1999 Aug; 19(16):RC20
Interdependence of multiple theta generators in the hippocampus: a partial coherence analysis. Kocsis B, Bragin A, Buzsaki G. J Neurosci 1999 Jul; 19(14):6200-12
Interactions between hippocampus and medial septum during sharp waves and theta oscillation in the behaving rat.
Dragoi G, Carpi D, Recce M, Csicsvari J, Buzsaki G. J Neurosci 1999 Jul; 19(14):6191-9
The entorhino-septo-supramammillary nucleus connection in the rat: morphological basis of a feedback mechanism regulating hippocampal theta rhythm. Leranth C, Carpi D, Buzsaki G, Kiss J. Neuroscience 1999; 88(3):701-18
High-frequency oscillations in human brain. Bragin A, Engel J Jr, Wilson CL, Fried I, Buzsaki G. Hippocampus 1999; 9(2):137-42
Sustained activation of hippocampal pyramidal cells by ‘space clamping’ in a running wheel. Czurko A, Hirase H, Csicsvari J, Buzsaki G. Eur J Neurosci 1999 Jan; 11(1):344-52
Oscillatory coupling of hippocampal pyramidal cells and interneurons in the behaving Rat. Csicsvari J, Hirase H, Czurko A, Mamiya A, Buzsaki G. J Neurosci 1999 Jan; 19(1):274-87
Gamma frequency oscillation in the hippocampus of the rat: intracellular analysis in vivo. Penttonen M, Kamondi A, Acsady L, Buzsaki G. Eur J Neurosci 1998 Feb; 10(2):718-28
Reliability and state dependence of pyramidal cell-interneuron synapses in the hippocampus: an ensemble approach in the behaving rat. Csicsvari J, Hirase H, Czurko A, Buzsaki G. Neuron 1998 Jul; 21(1):179-89
Remembering the Caribbean: the Spring Hippocampal Research Conference. Buhl EH, Buzsaki G. Neuron 1998 Jul; 21(1):27-35
Memory consolidation during sleep: a neurophysiological perspective. Buzsaki G. J Sleep Res 1998; 7 Suppl 1():17-23
Theta oscillations in somata and dendrites of hippocampal pyramidal cells in vivo: activity-dependent phase-precession of action potentials. Kamondi A, Acsady L, Wang XJ, Buzsaki G. Hippocampus 1998; 8(3):244-61
Dendritic spikes are enhanced by cooperative network activity in the intact hippocampus. Kamondi A, Acsady L, Buzsaki G. J Neurosci 1998 May; 18(10):3919-28
Induction of calcitonin gene-related peptide-like immunoreactivity in hippocampal neurons following ischemia: a putative regional modulator of the CNS injury/immune response. Bulloch K, Milner TA, Prasad A, Hsu M, Buzsaki G, McEwen BS. Exp Neurol 1998 Apr; 150(2):195-205
Operational dynamics in the hippocampal-entorhinal axis. Chrobak JJ, Buzsaki G. Neurosci Biobehav Rev 1998 Mar; 22(2):303-10
GABAergic cells are the major postsynaptic targets of mossy fibers in the rat hippocampus. Acsady L, Kamondi A, Sik A, Freund T, Buzsaki G. J Neurosci 1998 May; 18(9):3386-403
Somadendritic backpropagation of action potentials in cortical pyramidal cells of the awake rat. Buzsaki G, Kandel A. J Neurophysiol 1998 Mar; 79(3):1587-91
Dendritic properties of hippocampal CA1 pyramidal neurons in the rat: intracellular staining in vivo and in vitro. Pyapali GK, Sik A, Penttonen M, Buzsaki G, Turner DA. J Comp Neurol 1998 Feb; 391(3):335-52
Gamma oscillations in the entorhinal cortex of the freely behaving rat. Chrobak JJ, Buzsaki G. J Neurosci 1998 Jan; 18(1):388-98
Cellular-synaptic generation of sleep spindles, spike-and-wave discharges, and evoked thalamocortical responses in the neocortex of the rat. Kandel A, Buzsaki G. J Neurosci 1997 Sep; 17(17):6783-97
Functions for interneuronal nets in the hippocampus. Buzsaki G. Can J Physiol Pharmacol 1997 May; 75(5):508-15
MK-801-induced neuronal damage in rats. Horvath ZC, Czopf J, Buzsaki G. Brain Res 1997 Apr; 753(2):181-95
Termination of epileptic afterdischarge in the hippocampus. Bragin A, Penttonen M, Buzsaki G. J Neurosci 1997 Apr; 17(7):2567-79
Interneurons in the hippocampal dentate gyrus: an in vivo intracellular study. Sik A, Penttonen M, Buzsaki G. Eur J Neurosci 1997 Mar; 9(3):573-88
Epileptic afterdischarge in the hippocampal-entorhinal system: current source density and unit studies. Bragin A, Csicsvari J, Penttonen M, Buzsaki G. Neuroscience 1997 Feb; 76(4):1187-203
Feed-forward and feed-back activation of the dentate gyrus in vivo during dentate spikes and sharp wave bursts. Penttonen M, Kamondi A, Sik A, Acsady L, Buzsaki G. Hippocampus 1997; 7(4):437-50
Gamma oscillation by synaptic inhibition in a hippocampal interneuronal network model. Wang XJ, Buzsaki G. J Neurosci 1996 Oct; 16(20):6402-13
Pattern and inhibition-dependent invasion of pyramidal cell dendrites by fast spikes in the hippocampus in vivo. Buzsaki G, Penttonen M, Nadasdy Z, Bragin A. Proc Natl Acad Sci U S A 1996 Sep; 93(18):9921-5
Analysis of gamma rhythms in the rat hippocampus in vitro and in vivo. Traub RD, Whittington MA, Colling SB, Buzsaki G, Jefferys JG. J Physiol 1996 Jun; 493 ( Pt 2)():471-84
High-frequency oscillations in the output networks of the hippocampal-entorhinal axis of the freely behaving rat. Chrobak JJ, Buzsaki G. J Neurosci 1996 May; 16(9):3056-66
Lack of hippocampal involvement in a rat model of petit mal epilepsy. Kandel A, Bragin A, Carpi D, Buzsaki G. Epilepsy Res 1996 Mar; 23(2):123-7
The hippocampo-neocortical dialogue. Buzsaki G. Cereb Cortex 1996 Mar; 6(2):81-92
AMPA receptors in the rat and primate hippocampus: a possible absence of GluR2/3 subunits in most interneurons. Leranth C, Szeidemann Z, Hsu M, Buzsaki G. Neuroscience 1996 Feb; 70(3):631-52
Structural impairment of hippocampal neurons following a single epileptic afterdischarge. Horvath Z, Hsu M, Pierre E, Vadi D, Gallyas F, Buzsaki G. Epilepsy Res Suppl 1996; 12():325-34
Interneurons of the hippocampus. Freund TF, Buzsaki G. Hippocampus 1996; 6(4):347-470
Entorhinal cortical innervation of parvalbumin-containing neurons (Basket and Chandelier cells) in the rat Ammon’s horn. Kiss J, Buzsaki G, Morrow JS, Glantz SB, Leranth C. Hippocampus 1996; 6(3):239-46
GABAergic inhibition of granule cells and hilar neuronal synchrony following ischemia-induced hilar neuronal loss. Mody I, Otis TS, Bragin A, Hsu M, Buzsaki G. Neuroscience 1995 Nov; 69(1):139-50
Hippocampal CA1 interneurons: an in vivo intracellular labeling study. Sik A, Penttonen M, Ylinen A, Buzsaki G. J Neurosci 1995 Oct; 15(10):6651-65
Epileptic seizures caused by inactivation of a novel gene, jerky, related to centromere binding protein-B in transgenic mice. Toth M, Grimsby J, Buzsaki G, Donovan GP. Nat Genet 1995 Sep; 11(1):71-5
Temporal structure in spatially organized neuronal ensembles: a role for interneuronal networks. Buzsaki G, Chrobak JJ. Curr Opin Neurobiol 1995 Aug; 5(4):504-10
Morphometric and electrical properties of reconstructed hippocampal CA3 neurons recorded in vivo. Turner DA, Li XG, Pyapali GK, Ylinen A, Buzsaki G. J Comp Neurol 1995 Jun; 356(4):580-94
Effect of cerebral ischemia on calcium/calmodulin-dependent protein kinase II activity and phosphorylation. Shackelford DA, Yeh RY, Hsu M, Buzsaki G, Zivin JA. J Cereb Blood Flow Metab 1995 May; 15(3):450-61
Dentate EEG spikes and associated interneuronal population bursts in the hippocampal hilar region of the rat. Bragin A, Jando G, Nadasdy Z, van Landeghem M, Buzsaki G. J Neurophysiol 1995 Apr; 73(4):1691-705
Genetic threshold hypothesis of neocortical spike-and-wave discharges in the rat: an animal model of petit mal epilepsy. Vadasz C, Carpi D, Jando G, Kandel A, Urioste R, Horvath Z, Pierre E, Vadi D, Fleischer A, Buzsaki G. Am J Med Genet 1995 Feb; 60(1):55-63
Gamma (40-100 Hz) oscillation in the hippocampus of the behaving rat. Bragin A, Jando G, Nadasdy Z, Hetke J, Wise K, Buzsaki G. J Neurosci 1995 Jan; 15(1 Pt 1):47-60
Sharp wave-associated high-frequency oscillation (200 Hz) in the intact hippocampus: network and intracellular mechanisms. Ylinen A, Bragin A, Nadasdy Z, Jando G, Szabo I, Sik A, Buzsaki G. J Neurosci 1995 Jan; 15(1 Pt 1):30-46
Intracellular correlates of hippocampal theta rhythm in identified pyramidal cells, granule cells, and basket cells. Ylinen A, Soltesz I, Bragin A, Penttonen M, Sik A, Buzsaki G. Hippocampus 1995; 5(1):78-90
Spike-and-wave epilepsy in rats: sex differences and inheritance of physiological traits. Jando G, Carpi D, Kandel A, Urioste R, Horvath Z, Pierre E, Vadi D, Vadasz C, Buzsaki G. Neuroscience 1995 Jan; 64(2):301-17
Possible physiological role of the perforant path-CA1 projection. Buzsaki G, Penttonen M, Bragin A, Nadasdy Z, Chrobak JJ. Hippocampus 1995; 5(2):141-6
Short-term and long-term changes in the postischemic hippocampus. Hsu M, Sik A, Gallyas F, Horvath Z, Buzsaki G. Ann N Y Acad Sci 1994 Nov; 743():121-39; discussion 139-40
Selective activation of deep layer (V-VI) retrohippocampal cortical neurons during hippocampal sharp waves in the behaving rat. Chrobak JJ, Buzsaki G. J Neurosci 1994 Oct; 14(10):6160-70
Hippocampal theta activity following selective lesion of the septal cholinergic system. Lee MG, Chrobak JJ, Sik A, Wiley RG, Buzsaki G. Neuroscience 1994 Oct; 62(4):1033-47
Inhibitory CA1-CA3-hilar region feedback in the hippocampus. Sik A, Ylinen A, Penttonen M, Buzsaki G. Science 1994 Sep; 265(5179):1722-4
The hippocampal CA3 network: an in vivo intracellular labeling study. Li XG, Somogyi P, Ylinen A, Buzsaki G. J Comp Neurol 1994 Jan; 339(2):181-208
Four modified silver methods for thick sections of formaldehyde-fixed mammalian central nervous tissue: ‘dark’ neurons, perikarya of all neurons, microglial cells and capillaries. Gallyas F, Hsu M, Buzsaki G. J Neurosci Methods 1993 Nov; 50(2):159-64
Vulnerability of mossy fiber targets in the rat hippocampus to forebrain ischemia. Hsu M, Buzsaki G. J Neurosci 1993 Sep; 13(9):3964-79
Morphologic alterations of choline acetyltransferase-positive neurons in the basal forebrain of aged behaviorally characterized Fisher 344 rats.
Armstrong DM, Sheffield R, Buzsaki G, Chen KS, Hersh LB, Nearing B, Gage FH. Neurobiol Aging 1993 Sep; 14(5):457-70
Cerebellar neuronal activity correlates with spike and wave EEG patterns in the rat. Kandel A, Buzsaki G. Epilepsy Res 1993 Sep; 16(1):1-9
Pattern recognition of the electroencephalogram by artificial neural networks. Jando G, Siegel RM, Horvath Z, Buzsaki G. Electroencephalogr Clin Neurophysiol 1993 Feb; 86(2):100-9
Persisting axonal degeneration in the hippocampus after transection of the fimbria-fornix. Miettinen R, Lahtinen H, Riekkinen PJ, Freund TF, Hsu M, Horvath Z, Buzsaki G. Restor Neurol Neurosci 1993 Jan; 5(3):215-23
Delayed degeneration of the optic tract and neurons in the superior colliculus after forebrain ischemia. Gallyas F, Hsu M, Buzsaki G. Neurosci Lett 1992 Sep; 144(1-2):177-9
Intraseptal connections redefined: lack of a lateral septum to medial septum path. Leranth C, Deller T, Buzsaki G. Brain Res 1992 Jun; 583(1-2):1-11
High-frequency network oscillation in the hippocampus. Buzsaki G, Horvath Z, Urioste R, Hetke J, Wise K. Science 1992 May; 256(5059):1025-7
Alterations in [3H]kainate and N-methyl-D-aspartate-sensitive L-[3H]-glutamate binding in the rat hippocampal formation following fimbria-fornix lesions. Geddes JW, Brunner L, Cotman CW, Buzsaki G. Exp Neurol 1992 Feb; 115(2):271-81
Physiological and behavioral consequences of delayed septal grafts in the subcortically denervated hippocampus. Buzsaki G, Hsu M, Horvath Z, Gage FH. Restor Neurol Neurosci 1992 Jan; 4(6):369-80
Computer simulation of carbachol-driven rhythmic population oscillations in the CA3 region of the in vitro rat hippocampus. Traub RD, Miles R, Buzsaki G. J Physiol 1992; 451():653-72
Axonal and dendritic arborization of an intracellularly labeled chandelier cell in the CA1 region of rat hippocampus. Li XG, Somogyi P, Tepper JM, Buzsaki G. Exp Brain Res 1992; 90(3):519-25
Kindling-induced changes of protein kinase C levels in hippocampus and neocortex. Buzsaki G, Hsu M, Horvath Z, Horsburgh K, Sundsmo M, Masliah E, Saitoh T. Epilepsy Res Suppl 1992; 9():279-83; discussion 283-4
Physiological function of granule cells: a hypothesis. Buzsaki G, Czeh G. Epilepsy Res Suppl 1992; 7():281-90
Fetal brain tissue grafts modulate neuronal excitability in a chronic model of epilepsy. Buzsaki G, Gage FH. Epilepsy Res Suppl 1992; 8():271-81
Distributed changes in rat brain DNA synthesis with long-term habituation and potentiation of the perforant path-granule cell synapse. Sadile AG, Neugebauer A, Morelli F, Horvath Z, Buzsaki G, Giuditta A. Behav Brain Res 1991 Dec; 46(1):83-94
Network properties of memory trace formation in the hippocampus. Buzsaki G. Boll Soc Ital Biol Sper 1991 Sep; 67(9):817-35
Hippocampal grafts into the intact brain induce epileptic patterns. Buzsaki G, Masliah E, Chen LS, Horvath Z, Terry R, Gage FH. Brain Res 1991 Jul; 554(1-2):30-7
The path forward in Hungarian neuroscience. Antal M, Buzsaki G, Czeh G, Gaal G, Galyas B, Horvath Z, Jakab R, Kabai P, Kiss J, Kocsis B, et al.. Trends Neurosci 1991 May; 14(5):185-8
Emergence and propagation of interictal spikes in the subcortically denervated hippocampus. Buzsaki G, Hsu M, Slamka C, Gage FH, Horvath Z. Hippocampus 1991 Apr; 1(2):163-80
The cholinergic system and EEG slow waves. Riekkinen P, Buzsaki G, Riekkinen P Jr, Soininen H, Partanen J. Electroencephalogr Clin Neurophysiol 1991 Feb; 78(2):89-96
Noradrenergic Control of Thalamic Oscillation: the Role of alpha-2 Receptors. Buzsaki G, Kennedy B, Solt VB, Ziegler M. Eur J Neurosci 1991; 3(3):222-229
The thalamic clock: emergent network properties. Buzsaki G. Neuroscience 1991; 41(2-3):351-64
Hippocampal cell death following ischemia: effects of brain temperature and anesthesia. Freund TF, Buzsaki G, Leon A, Somogyi P. Exp Neurol 1990 Jun; 108(3):251-60
Nucleus basalis lesions fail to induce senile plaques in the rat. Thal LJ, Mandel RJ, Terry RD, Buzsaki G, Gage FH. Exp Neurol 1990 Apr; 108(1):88-90
Coherence of compound field potentials reveals discontinuities in the CA1-subiculum of the hippocampus in freely-moving rats. Bullock TH, Buzsaki G, McClune MC. Neuroscience 1990; 38(3):609-19
Spike-and-wave neocortical patterns in rats: genetic and aminergic control. Buzsaki G, Laszlovszky I, Lajtha A, Vadasz C. Neuroscience 1990; 38(2):323-33
NGF-dependent sprouting and regeneration in the hippocampus. Gage FH, Buzsaki G, Armstrong DM. Prog Brain Res 1990; 83():357-70
Spatial organization of physiological activity in the hippocampal region: relevance to memory formation. Buzsaki G, Chen LS, Gage FH. Prog Brain Res 1990; 83():257-68
Petit mal epilepsy and parkinsonian tremor: hypothesis of a common pacemaker. Buzsaki G, Smith A, Berger S, Fisher LJ, Gage FH. Neuroscience 1990; 36(1):1-14
Relationship of neuronal vulnerability and calcium binding protein immunoreactivity in ischemia. Freund TF, Buzsaki G, Leon A, Baimbridge KG, Somogyi P. Exp Brain Res 1990; 83(1):55-66
Nerve growth factor infusions combined with fetal hippocampal grafts enhance reconstruction of the lesioned septohippocampal projection. Tuszynski MH, Buzsaki G, Gage FH. Neuroscience 1990; 36(1):33-44
Thalamic nuclei in Alzheimer disease: evidence against the cholinergic hypothesis of plaque formation. Masliah E, Terry R, Buzsaki G. Brain Res 1989 Jul; 493(2):240-6
The grafted hippocampus: an epileptic focus. Buzsaki G, Bayardo F, Miles R, Wong RK, Gage FH. Exp Neurol 1989 Jul; 105(1):10-22
Multisite recording of brain field potentials and unit activity in freely moving rats. Buzsaki G, Bickford RG, Ryan LJ, Young S, Prohaska O, Mandel RJ, Gage FH. J Neurosci Methods 1989 Jun; 28(3):209-17
Absence of long-term potentiation in the subcortically deafferented dentate gyrus. Buzsaki G, Gage FH. Brain Res 1989 Apr; 484(1-2):94-101
The effects of unilateral destruction of fimbria-fornix and supracallosal pathways in the rat. Buzsaki G, Ryan JP, Isaacson RL. Behav Neural Biol 1989 Mar; 51(2):278-88
Long-term potentiation of evoked and spontaneous neuronal activity in the grafted hippocampus. Buzsaki G, Wiesner J, Henriksen SJ, Gage FH. Exp Brain Res 1989; 76(2):401-8
Simultaneous recording of local electrical activity, partial oxygen tension and temperature in the rat hippocampus with a chamber-type microelectrode. Effects of anaesthesia, ischemia and epilepsy. Freund TF, Buzsaki G, Prohaska OJ, Leon A, Somogyi P. Neuroscience 1989; 28(3):539-49
Neuronal activity in the subcortically denervated hippocampus: a chronic model for epilepsy. Buzsaki G, Ponomareff GL, Bayardo F, Ruiz R, Gage FH. Neuroscience 1989; 28(3):527-38
Two-stage model of memory trace formation: a role for “noisy” brain states. Buzsaki G. Neuroscience 1989; 31(3):551-70
[PDF]
Ischemia-induced changes in the electrical activity of the hippocampus. Buzsaki G, Freund TF, Bayardo F, Somogyi P. Exp Brain Res 1989; 78(2):268-78
Survival, growth and function of damaged cholinergic neurons. Gage FH, Tuszynski MH, Chen KS, Armstrong D, Buzsaki G. EXS 1989; 57():259-74
The cholinergic nucleus basalis: a key structure in neocortical arousal. Buzsaki G, Gage FH. EXS 1989; 57():159-71
Suppression and induction of epileptic activity by neuronal grafts. Buzsaki G, Ponomareff G, Bayardo F, Shaw T, Gage FH. Proc Natl Acad Sci U S A 1988 Dec; 85(23):9327-30
Alterations in excitatory and GABAergic inhibitory connections in hippocampal transplants. Freund TF, Buzsaki G. Neuroscience 1988 Nov; 27(2):373-85
Nucleus basalis and thalamic control of neocortical activity in the freely moving rat. Buzsaki G, Bickford RG, Ponomareff G, Thal LJ, Mandel R, Gage FH. J Neurosci 1988 Nov; 8(11):4007-26
Electric activity in the neocortex of freely moving young and aged rats. Buzsaki G, Bickford RG, Armstrong DM, Ponomareff G, Chen KS, Ruiz R, Thal LJ, Gage FH. Neuroscience 1988 Sep; 26(3):735-44
Experimental approaches to age-related cognitive impairments. Gage FH, Chen KS, Buzsaki G, Armstrong D. Neurobiol Aging 1988 Sep; 9(5-6):645-55
Polysynaptic long-term potentiation: a physiological role of the perforant path–CA3/CA1 pyramidal cell synapse. Buzsaki G. Brain Res 1988 Jul; 455(1):192-5
Neocortical and hippocampal electrical activity following decapitation in the rat. Vanderwolf CH, Buzsaki G, Cain DP, Cooley RK, Robertson B. Brain Res 1988 Jun; 451(1-2):340-4
Mechanisms of action of neural grafts in the limbic system. Buzsaki G, Gage FH. Can J Neurol Sci 1988 May; 15(2):99-105
Perforant path activation of the hippocampus: spatial distribution, effects of urethane and atropine. Kamondi A, Horvath Z, Bors L, Buzsaki G. Acta Physiol Hung 1988; 71(1):19-29
Restoration and deterioration of function by brain grafts in the septohippocampal system. Buzsaki G, Freund T, Bjorklund A, Gage FH. Prog Brain Res 1988; 78():69-77
Long-term potentiation induced by physiologically relevant stimulus patterns. Buzsaki G, Haas HL, Anderson EG. Brain Res 1987 Dec; 435(1-2):331-3
Cellular activity of intracerebrally transplanted fetal hippocampus during behavior. Buzsaki G, Czopf J, Kondakor I, Bjorklund A, Gage FH. Neuroscience 1987 Sep; 22(3):871-83
Restoration of rhythmic slow activity (theta) in the subcortically denervated hippocampus by fetal CNS transplants. Buzsaki G, Gage FH, Czopf J, Bjorklund A. Brain Res 1987 Jan; 400(2):334-47
Behavioral dependence of the electrical activity of intracerebrally transplanted fetal hippocampus. Buzsaki G, Gage FH, Kellenyi L, Bjorklund A. Brain Res 1987 Jan; 400(2):321-33
Grafts of fetal cholinergic neurons to the deafferented hippocampus. Gage FH, Buzsaki G, Nilsson O, Bjorklund A. Prog Brain Res 1987; 71():335-47
Application of the glycine labelling method to the cerebellum, hippocampus and spinal cord. Rojik I, Baranyi A, Buzsaki G, Urban L, Feher O. Acta Biol Hung 1987; 38(3-4):393-407
Septal deafferentation produces continuous rhythmic slow activity (theta) in the rat hippocampus. Kvirkvelia L, Buzsaki G, Grastyan E. Acta Physiol Hung 1987; 70(1):127-31
Hippocampal sharp waves: their origin and significance. Buzsaki G. Brain Res 1986 Nov; 398(2):242-52
Hippocampal responses evoked by tooth pulp and acoustic stimulation: depth profiles and effect of behavior. Brankack J, Buzsaki G. Brain Res 1986 Jul; 378(2):303-14
Effects of confinement, previous experience and hippocampal damage on the DRL schedule. Acsadi G, Buzsaki G, Keszthelyi T, Kiralyfalvi L, Gage FH. Behav Brain Res 1986 May; 20(2):241-8
Laminar distribution of hippocampal rhythmic slow activity (RSA) in the behaving rat: current-source density analysis, effects of urethane and atropine. Buzsaki G, Czopf J, Kondakor I, Kellenyi L. Brain Res 1986 Feb; 365(1):125-37
Depth profiles of hippocampal rhythmic slow activity (‘theta rhythm’) depend on behaviour. Buzsaki G, Rappelsberger P, Kellenyi L. Electroencephalogr Clin Neurophysiol 1985 Jul; 61(1):77-88
Long-term changes of hippocampal sharp-waves following high frequency afferent activation. Buzsaki G. Brain Res 1984 May; 300(1):179-82
Feed-forward inhibition in the hippocampal formation. Buzsaki G. Prog Neurobiol 1984; 22(2):131-53
[PDF]
Spectral analysis of hippocampal unit train in relation to hippocampal EEG. Leung LW, Buzsaki G. Electroencephalogr Clin Neurophysiol 1983 Dec; 56(6):668-71
Cellular bases of hippocampal EEG in the behaving rat. Buzsaki G, Leung LW, Vanderwolf CH. Brain Res 1983 Oct; 287(2):139-71
Phase relations of hippocampal projection cells and interneurons to theta activity in the anesthetized rat. Buzsaki G, Eidelberg E. Brain Res 1983 May; 266(2):334-9
Situational conditional reflexes. Physiologic studies of the higher nervous activity of freely moving animals: P. S. Kupalov. Buzsaki G. Pavlov J Biol Sci 1983 Jan; 18(1):13-21
Direct afferent excitation and long-term potentiation of hippocampal interneurons. Buzsaki G, Eidelberg E. J Neurophysiol 1982 Sep; 48(3):597-607
The “where is it?” reflex: autoshaping the orienting response. Buzsaki G. J Exp Anal Behav 1982 May; 37(3):461-84
[PDF]
Convergence of associational and commissural pathways on CA1 pyramidal cells of the rat hippocampus. Buzsaki G, Eidelberg E. Brain Res 1982 Apr; 237(2):283-95
Spatial mapping, working memory, and the fimbria-fornix system. Buzsaki G, Bors L, Nagy F, Eidelberg E. J Comp Physiol Psychol 1982 Feb; 96(1):26-34
Commissural projection to the dentate gyrus of the rat: evidence for feed-forward inhibition. Buzsaki G, Eidelberg E. Brain Res 1981 Dec; 230(1-2):346-50
Changes in neuronal transmission in the rat hippocampus during behavior. Buzsaki G, Grastyan E, Czopf J, Kellenyi L, Prohaska O. Brain Res 1981 Nov; 225(2):235-47
Hippocampal slow wave activity during appetitive and aversive conditioning in the cat. Buzsaki G, Haubenreiser J, Grastyan E, Czopf J, Kellenyi L. Electroencephalogr Clin Neurophysiol 1981 Mar; 51(3):276-90
Commissural and perforant path interactions in the rat hippocampus. Field potentials and unitary activity. Buzsaki G, Czeh G. Exp Brain Res 1981; 43(3-4):429-38
Long-term potentiation of the commissural path-CA1 pyramidal cell synapse in the hippocampus of the freely moving rat. Buzsaki G. Neurosci Lett 1980 Oct; 19(3):293-6
Importance of cue location for intact and fimbria-fornix-lesioned rats. Buzsaki G, Grastyan E, Mod L, Winiczai Z. Behav Neural Biol 1980 Jun; 29(2):176-89
Effects of scopolamine upon hippocampal electrical activity associated with running and swimming in rats. Buzsaki G, Kellenyi L, Grastyan E. Physiol Behav 1980 Jan; 24(1):191-4
Differential contribution of fimbria and fornix fibers to behavior. Buzsaki G, Acsadi G, Jani A. Behav Neural Biol 1980 Jan; 28(1):79-88
Hippocampal evoked potentials and EEG changes during classical conditioning in the rat. Buzsaki G, Grastyan E, Tveritskaya IN, Czopf J. Electroencephalogr Clin Neurophysiol 1979 Jul; 47(1):64-74
The orienting-exploratory response hypothesis of discriminative conditioning. Grastyan E, Buzsaki G. Acta Neurobiol Exp (Wars) 1979; 39(6):491-501
Maintenance of signal directed behavior in a response dependent paradigm: a systems approach. Buzsaki G, Grastyan E, Winiczai Z, Mod L. Acta Neurobiol Exp (Wars) 1979; 39(4):201-17
Auto-shaping or orienting? Buzsaki G, Grastyan E, Molnar P, Tveritskaya IN, Haubenreiser J. Acta Neurobiol Exp (Wars) 1979; 39(4):179-200
Dynamic phase-shifts between theta generators in the rat hippocampus. Buzsaki G, Grastyan E, Kellenyi L, Czopf J. Acta Physiol Acad Sci Hung 1979; 53(1):41-5
Simple device for the tape recording of complex behavioral situation on one track. Kellenyi L, Buzsaki G. Physiol Behav 1977 Sep; 19(3):449-50
[Electrophysiological analysis of the effect of atropine on the self-stimulation reaction in cats]. Baklavadzhian O, Buzsaki G, Angyan L. Acta Physiol Acad Sci Hung 1973; 44(3-4):217-26